Advertisements
Advertisements
प्रश्न
Give an example and suggest a reason for the following feature of the transition metal chemistry:
The lowest oxide of transition metal is basic, the highest is amphoteric/acidic.
Account for the following:
The lowest oxide of transition metal is basic whereas the highest oxide is amphoteric or acidic.
Account for the following:
The lowest oxide of transition metal is basic, and the highest is acidic.
उत्तर १
The lowest oxide has the lowest oxidation state of the transition metal. Hence, the oxide is basic and tends to react with acids to get oxidised to attain a higher oxidation state. Whereas the highest oxides are formed in higher oxidation states. As a result, these oxides are amphoteric acidic. For example, \[\ce{MnO}\] is basic, whereas \[\ce{Mn2O7}\] is acidic.
उत्तर २
In the case of a lower oxide of a transition metal, the metal atom has some electrons present in the valence shell of the metal atom that are not involved in bonding. As a result, it can donate electrons and behave as a base, whereas in the higher oxide of a transition metal, the metal atom does not have an electron in the valence shell for donation. As a result, it can accept electrons and behave as an acid.
APPEARS IN
संबंधित प्रश्न
Complete the following equations:
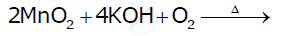
Complete the following equations: Cr2O72- + 14H+ + 6I →
Complete the following equation : MnO4- + 8H+ + 5e- →
Describe the preparation of potassium permanganate. How does the acidified permanganate solution react with iron (II) ions? Write the ionic equation for the reaction.
Give an example and suggest a reason for the following feature of the transition metal chemistry:
The highest oxidation state is exhibited in oxoanions of a metal.
Indicate the steps in the preparation of KMnO4 from pyrolusite ore.
Account for the following :
Manganese shows maximum number of oxidation states in 3d series.
Zinc carbonate is precipitated from zinc sulphate solution by the addition of ___________.
Why \[\ce{HCl}\] should not be used for potassium permanganate titrations?
What is the effect of pH on dichromate ion solution?
Potassium dichromate when heated with concentrated sulphuric acid and a soluble chloride, gives brown-red vapours of ______.
In the two tetrahedral structures of dichromate ion, ______.
Complete the reaction mentioning all the products formed:
\[\ce{2KMnO4 ->[\Delta]}\]
Indicate the steps in the preparation of K2Cr2O7 from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
