Advertisements
Advertisements
प्रश्न
A vessel of volume 125 cm3 contains tritium (3H, t1/2 = 12.3 y) at 500 kPa and 300 K. Calculate the activity of the gas.
उत्तर
Given:-
Volume of the vessel, V = 125 cm3 = 0.125 L
Half-life time of tritium, t1/2 = 12.3 y = 3.82 × 108 s
Pressure, P = 500 kpa = 5 atm
Temperature, T = 300 K,
Disintegration constant, `lambda = 0.693/t_"1/2"`
`0.693/(3.82 xx 10^8) = 0.1814 xx 10^-8`
`= 1.81 xx 10^-9 "s"^-1`
No. of atoms left undecayed, N = n × 6.023 × 1023
`= (5 xx 0.125)/(8.2 xx 10^-2 xx 3 xx 10^2) xx 6.023 xx 10^23...............(because n = "PV"/"RT")`
`= 1.5 xx 10^22` atoms
Activity, A = λN
`= 1.81 xx 10^-9 xx 1.5 xx 10^22 = 2.7 xx 10^13 " disintegration/sec"`
`therefore A = (2.7 xx 10^13)/(3.7 xx 10^10) "Ci" = 729.72` `"Ci"`
APPEARS IN
संबंधित प्रश्न
For the past some time, Aarti had been observing some erratic body movement, unsteadiness and lack of coordination in the activities of her sister Radha, who also used to complain of severe headache occasionally. Aarti suggested to her parents to get a medical check-up of Radha. The doctor thoroughly examined Radha and diagnosed that she has a brain tumour.
(a) What, according to you, are the values displayed by Aarti?
(b) How can radioisotopes help a doctor to diagnose brain tumour?
Write nuclear reaction equation for α-decay of `""_94^242"Pu"`.
Write nuclear reaction equation for β−-decay of `""_15^32"P"`.
Write nuclear reaction equation for β−-decay of `""_83^210"Bi"`.
Write nuclear reaction equation for β+-decay of `""_6^11"C"`.
The sequence of stepwise decay of a radioactive nucleus is
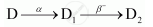
If the atomic number and mass number of D2 are 71 and 176 respectively, what are their corresponding values of D?
A certain sample of a radioactive material decays at the rate of 500 per second at a certain time. The count rate falls to 200 per second after 50 minutes. (a) What is the decay constant of the sample? (b) What is its half-life?
The count rate from a radioactive sample falls from 4.0 × 106 per second to 1.0 × 106per second in 20 hours. What will be the count rate 100 hours after the beginning?
The selling rate of a radioactive isotope is decided by its activity. What will be the second-hand rate of a one month old 32P(t1/2 = 14.3 days) source if it was originally purchased for 800 rupees?
`""_80^197`Hg decay to `""_79^197`Au through electron capture with a decay constant of 0.257 per day. (a) What other particle or particles are emitted in the decay? (b) Assume that the electron is captured from the K shell. Use Moseley's law √v = a(Z − b) with a = 4.95 × 107s−1/2 and b = 1 to find the wavelength of the Kα X-ray emitted following the electron capture.
The count rate of nuclear radiation coming from a radiation coming from a radioactive sample containing 128I varies with time as follows.
| Time t (minute): | 0 | 25 | 50 | 75 | 100 |
| Ctount rate R (109 s−1): | 30 | 16 | 8.0 | 3.8 | 2.0 |
(a) Plot In (R0/R) against t. (b) From the slope of the best straight line through the points, find the decay constant λ. (c) Calculate the half-life t1/2.
Natural water contains a small amount of tritium (`""_1^3H`). This isotope beta-decays with a half-life of 12.5 years. A mountaineer while climbing towards a difficult peak finds debris of some earlier unsuccessful attempt. Among other things he finds a sealed bottled of whisky. On returning, he analyses the whisky and finds that it contains only 1.5 per cent of the `""_1^3H` radioactivity as compared to a recently purchased bottle marked '8 years old'. Estimate the time of that unsuccessful attempt.
4 × 1023 tritium atoms are contained in a vessel. The half-life of decay tritium nuclei is 12.3 y. Find (a) the activity of the sample, (b) the number of decay in the next 10 hours (c) the number of decays in the next 6.15 y.
238U decays to 206Pb with a half-life of 4.47 × 109 y. This happens in a number of steps. Can you justify a single half for this chain of processes? A sample of rock is found to contain 2.00 mg of 238U and 0.600 mg of 206Pb. Assuming that all the lead has come from uranium, find the life of the rock.
In a gamma ray emission from nucleus :
A radioactive substance decays to 1/16th of its initial mass in 40 days. The half-life of the substance, in days, is:
The half-life of a certain radioactive element is 3.465 days. Find its disintegration constant.
