Advertisements
Advertisements
प्रश्न
Account for the following:
Acylation of aniline is carried out in the presence of pyridine.
उत्तर
During the acylation of aniline, stronger base pyridine is added. This done in order to remove the HCl so formed during the reaction and to shift the equilibrium to the right hand side.
APPEARS IN
संबंधित प्रश्न
Write the chemical equations involved when aniline is treated with the following reagents: HCI
Why does NH3 act as a Lewis base?
Arrange the following:
In decreasing order of basic strength in the gas phase:
C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3
The correct increasing order of basic strength for the following compounds is ______.
(I)
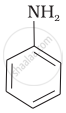
(II)
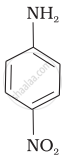
(III)
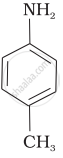
The most reactive amine towards dilute hydrochloric acid is:
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
When methyl iodide is heated with ammonia, what is the product obtained?
What is the characteristic smell of liquid amines?
Which of the following compound cannot be produced if 1-propane amine is treated with NaNO2 and HCl?
Arrange the decreasing order of pKb values.
\[\ce{C6H5NH2, C6H5NHCH3, C6H5CH2NH2, CH3NH2, NH3}\]
