Advertisements
Advertisements
प्रश्न
The correct increasing order of basic strength for the following compounds is ______.
(I)
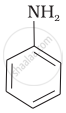
(II)
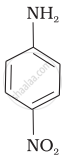
(III)
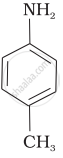
पर्याय
II < III < I
III < I < II
III < II < I
II < I < III
उत्तर
The correct increasing order of basic strength for the following compounds is II < I < III.
Explanation:
The greater the electron density towards the ring, the greater its basic strength. The electron-withdrawing group reduces basic strength, whereas the electron-donating group increases basic strength.
APPEARS IN
संबंधित प्रश्न
Write the structures of main products when aniline reacts with the following reagents : HCl
Write the chemical equations involved when aniline is treated with the following reagents: HCI
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5NHCH3, C6H5CH2NH2
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
When ethanol is mixed with ammonia and passed over alumina the compound formed is which compound?
Which of the following is most basic?
What is the correct decreasing order of the basic character of the three amines and ammonia?
Arrange the decreasing order of pKb values.
\[\ce{C6H5NH2, C6H5NHCH3, C6H5CH2NH2, CH3NH2, NH3}\]
State one reason for the following:
Alkylamine is soluble in water, whereas arylamine is insoluble in water.
