Advertisements
Advertisements
प्रश्न
Assertion: Order of the reaction can be zero or fractional.
Reason: We cannot determine order from balanced chemical equation.
पर्याय
Both assertion and reason are correct and the reason is correct explanation of assertion.
Both assertion and reason are correct but reason does not explain assertion.
Assertion is correct but reason is incorrect.
Both assertion and reason are incorrect.
Assertion is incorrect but reason is correct.
उत्तर
Both assertion and reason are correct but reason does not explain assertion.
Explanation:
Order of a reaction may be zero or fractional. Order can be determined by rate law expression.
APPEARS IN
संबंधित प्रश्न
In a first order reaction x → y, 40% of the given sample of compound remains unreacted in 45 minutes. Calculate rate constant of the reaction.
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For the first order thermal decomposition reaction, the following data were obtained:
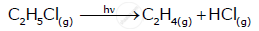
Time / sec Totalpressure / atm
0 0.30
300 0.50
Calculate the rate constant
(Given: log 2 = 0.301, log3 = 0.4771, log 4 = 0.6021)
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
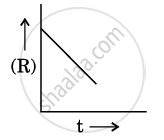
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
A reaction is first order in A and second order in B. How is the rate affected when the concentrations of both A and B are doubled?
In a reaction if the concentration of reactant A is tripled, the rate of reaction becomes twenty seven times. What is the order of the reaction?
Assertion: The enthalpy of reaction remains constant in the presence of a catalyst.
Reason: A catalyst participating in the reaction, forms different activated complex and lowers down the activation energy but the difference in energy of reactant and product remains the same.
Assertion (A): Order of reaction is applicable to elementary as well as complex reactions.
Reason (R): For a complex reaction, molecularity has no meaning.
Which of the following statement is true?
