Advertisements
Advertisements
प्रश्न
Compound ‘A’ with molecular formula \[\ce{C4H9Br}\] is treated with aq. \[\ce{KOH}\] solution. The rate of this reaction depends upon the concentration of the compound ‘A’ only. When another optically active isomer ‘B’ of this compound was treated with aq. \[\ce{KOH}\] solution, the rate of reaction was found to be dependent on concentration of compound and \[\ce{KOH}\] both.
(i) Write down the structural formula of both compounds ‘A’ and ‘B’.
(ii) Out of these two compounds, which one will be converted to the product with inverted configuration.
उत्तर
(i) The molecular formulae of isomers of \[\ce{C4H9Br}\] are \[\ce{CH3}\]
\[\begin{array}{cc}
\ce{CH3}\phantom{......}\\
|\phantom{.........}\\
\ce{CH3 - C - Br}\phantom{...........}\\
|\phantom{.........}\\
\ce{\underset{2 - Bromo-2-methyl propane (A)}{CH3}}\phantom{......}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\phantom{}\\
\phantom{.......}\ce{\underset{2-Bromobutance (B)}{Br}}\phantom{}
\end{array}\]
Since the rate of reaction of compound ‘A’ \[\ce{(C4H9Br)}\] with aqueous \[\ce{KOH}\] depends upon the concentration of compound ‘A’ only, therefore, the reaction occurs by SN1 mechanism and compound ‘A’ is tertiary bromide i.e., 2-Bromo-2-methylpropane.
\[\ce{(CH3)3CBr + KOH(aq) –> (CH3)3COH + KBr}\]
Rate = \[\ce{A:[(CH3)3CBr]}\]
(ii) Since compound ‘B’ is optically active and is an isomer of compound ‘A’ \[\ce{(C4H9Br)}\], therefore, compound ‘B’ must be 2-Bromobutane. Since the rate of reaction of compound ‘B’ with aqueous \[\ce{KOH}\] depends upon the concentration of compound ‘B’ and \[\ce{KOH}\], therefore, the reaction occurs by SN2 mechanism and product of hydrolysis will have inverted configuration.
\[\begin{array}{cc}
\ce{CH3CH2CHCH3 + KOH -> CH3CH2CHCH3 + KBr}\\
\phantom{..}|\phantom{...............................}|\phantom{...}\\
\phantom{..}\ce{Br}\phantom{.............................}\ce{OH}\phantom{.}
\end{array}\]
Rate = 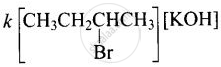
APPEARS IN
संबंधित प्रश्न
Which would undergo SN1 reaction faster in the following pair and why?
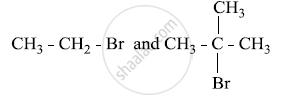
Give reasons for the following:
(CH3)3C–O–CH3 on reaction with HI gives (CH3)3C–I and CH3–OH as the main products and not (CH3)3C–OH and CH3–I.
SN1 reactions are accompanied by racemization in optically active alkyl halides.
Given reasons: SN1 reactions are accompanied by racemization in optically active alkyl halides.
What is the action of the following on ethyl bromide?
moist silver oxide
What is the action of the following on ethyl bromide:
silver acetate
Which of the following is the correct order of decreasing SN2 reactivity?
SN1 reaction of alkyl halides lead to ___________.
In which reaction mechanism carbocation is formed?
Retention of configuration is observed in ______.
