Advertisements
Advertisements
प्रश्न
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits ____________ isomerism
पर्याय
Coordination
Ionisation
Linkage
Optical
उत्तर
The pair [Co(NH3)5(SO4)]Br and [Co(NH3)5Br]SO4 exhibits Ionisation isomerism
APPEARS IN
संबंधित प्रश्न
Answer in brief.
What are ionization isomers ? Give an example.
Answer the following question.
Draw geometric isomers and enantiomers of the following complex.
[Pt(en)2ClBr]2⊕
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Define the term Co-ordination isomer.
Draw optical isomers of [Co(en)3]3+.
Which one of the following complexes is not expected to exhibit isomerism?
What is linkage isomerism? Explain with an example.
What are hydrate isomers? Explain with an example.
The term anomers of glucose refer to ____________.
Which would exhibit coordination isomerism?
Consider the two complexes given below:
\[\ce{\underset{(I)}{[Co(NH3)5SO4]Br}}\] and \[\ce{\underset{(II)}{[Co(NH3)5Br]SO4}}\]
I and II are ____________ isomers.
How many donor groups are present in diethylene triamine?
The relationship between compound (i) and (ii) is
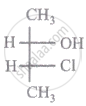 |
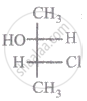 |
| (i) | (ii) |
Which of the following compound show optical isomerism?
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
Write the name of isomerism in the following complexes:
[Cu(NH3)4] [PtCl4] and [Pt(NH3)4] [ CuCl4]
Draw geometric isomers of the following complex.
Geometrical isomers of Pt(NH3)2Cl2
Which compound would exhibit optical isomers?
The number of geometrical isomers of \[\ce{[Co(NH3)3 (NO3)3]}\] are ______.
The compounds [PtCl2(NH3)4]Br2 and [PtBr2(NH3)4]Cl2 constitutes a pair of ______.
Give cis isomer of [Co(NH3)4Cl2]⊕.
Give trans isomer of [Co(NH3)4Cl2]⊕.
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Explain the ionisation isomers.
Draw the structure of cis isomers of Pt(NH3)2Cl2.
Name the type of isomerism exhibited by the following pair of compound:
\[\ce{[Co(NH3)5 [ONO]Cl2 and [Co(NH3)5(NO2)]Cl2}\]
Which one of the following complex ions has geometrical isomers?
