Advertisements
Advertisements
प्रश्न
The X-ray coming from a Coolidge tube has a cutoff wavelength of 80 pm. Find the kinetic energy of the electrons hitting the target.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
उत्तर
Given:-
Cutoff wavelength of the Coolidge tube, `lambda = 80 "pm"`
Energy of the electron hitting the target (E) is given by
`E = (hc)/lambda`
Here,
h = Planck's constant
c = Speed of light
λ = Wavelength of light
`therefore E = (1242 "eV"-"nm")/(80 xx 10^-3)`
`⇒ E = (1242 xx 10^-9 "eV")/(80 xx 10^-12)`
`⇒ E = 15.525 xx 10^3 "eV" ≈ 15.5 "keV"`
APPEARS IN
संबंधित प्रश्न
To which part of the electromagnetic spectrum does a wave of frequency 5 × 1019 Hz belong?
(a) Give a list of at least five radiations, in order of their increasing frequencies, which make up the complete electromagnetic spectrum.
(b) Which of the radiation mentioned by you in part (a) has the highest penetrating power.
What is the range of the wavelength of the following electromagnetic waves?
(a) Visible.
What are ultraviolet radiations?
Can X-rays be polarised?
Can Lα X-ray of one material have shorter wavelength than Kα X-ray of another?
An X-ray beam can be deflected
X-ray from a Coolidge tube is incident on a thin aluminium foil. The intensity of the X-ray transmitted by the foil is found to be I0. The heating current is increased to increase the temperature of the filament. The intensity of the X-ray transmitted by the foil will be
(a) zero
(b) < I0
(c) I0
(d) > I0
Find the energy, the frequency and the momentum of an X-ray photon of wavelength 0.10 nm.
(Use Planck constant h = 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The Kβ X-ray of argon has a wavelength of 0.36 nm. The minimum energy needed to ionize an argon atom is 16 eV. Find the energy needed to knock out an electron from the K shell of an argon atom.
Name the scientist who discovered Ultraviolet rays
Name three radiations and their wavelength range which are invisible and beyond the violet end of the visible spectrum.
To which regions of the electromagnetic spectrum do the following wavelengths belong:
(a) 250 nm
(b) 1500 nm
Answer briefly.
Why high-frequency carrier waves are used for the transmission of audio signals?
The electric field intensity produced by the radiations coming from 100 W bulb at a 3 m distance is E. The electric field intensity produced by the radiations coming from 50 W bulb at the same distance is ______.
In uranium (Z = 92) the K absorption edge is 0.107 Å and the Kα line is 0.126 Å, and the wavelength of the L absorption edge is ______.
What is the speed of radio waves in vacuum?
Name the electromagnetic radiation that has been used in obtaining the image below.
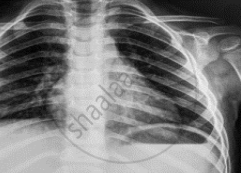
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
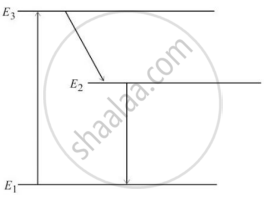
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
