Advertisements
Advertisements
प्रश्न
What is a cell constant? What are its units? How is it determined experimentally?
उत्तर
For a given cell, the ratio of separation (l) between the two electrodes divided by the area of cross-section (a) of the electrode is called the cell constant.
Cell constant = `"l"/"a"`
The unit of cell constant is m-1 (SI unit) or cm-1 (C.G.S unit).
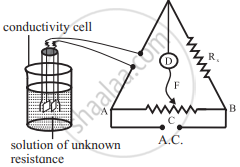
1) The cell constant is determined using the 1 M, 0.1 M or 0.01 M KCl solutions. The conductivity of KCl solution is well tabulated at various temperatures.
2) The resistance of KCl solution is measured by Wheatstone bridge as shown in the figure.
3) AB is the uniform wire. Rx is the variable known resistance placed in one arm of Wheatstone bridge. The conductivity cell containing KCl solution of unknown resistance is placed in the other arm of Wheatstone bridge.
4) D is a current detector. F is the sliding contact that moves along AB. A.C. represents the source of alternating current.
5) The sliding contact is moved along AB until no current flows. The detector D shows no deflection. The null point is, thus, obtained at C.
6) According to Wheatstone bridge principle,
`("R"_(("solution")))/(l("AC")) = "R"_"x"/(l("BC"))`
Hence, `"R"_(("solution")) = (l("AC"))/(l("BC")) xx "R"_"x"`
7) By measuring lengths AC and BC and knowing Rx , resistance of KCl solution can be calculated.
8) The cell constant is given by
Cell constant = kKCl × `"R"_(("solution"))`
The conductivity of KCl solution is known. The cell constant, thus, can be calculated.
संबंधित प्रश्न
Answer the following in brief.
Explain the effect of dilution of the solution on conductivity.
Kohlrausch law is applicable for the solution _______.
Write an equation that shows the relationship between molar conductivity and degree of dissociation of weak electrolyte.
Write applications of Kohlrausch’s Law.
Write three important steps required to determine molar conductivity.
Calculate molar conductivities at zero concentration for CaCl2 and NaCl.
Given: molar ionic conductivities of Ca2+, Cl–, Na+ ions are respectively, 104, 76.4, 50.1 Ω–1 cm–2 mol–1
Write relation between electrolytic conductivity and molar conductivity.
If resistivity of 0.8 M KCI solution is 2.5 × 10-3 Ω cm. Calculate molar conductivity of solution?
For the reaction \[\ce{A -> B}\], the rate increases by a factor of 2.25 when the concentration of A is increased by 1.5. What is the order of the reaction?
What will be the concentration of NaCl solution, if the molar conductivity and conductivity of NaCl solution is 124.3 Ω−1 cm2 mol−1 and 1.243 × 10−4 Ω−1 cm2 respectively?
A conductivity cell having cell constant 8.76 cm−1, placed in 0.01 M solution of an electrolyte offered a resistance of 1000 ohms. What is the specific conductance of electrolytes?
The SI unit of resistivity and molar conductivity are respectively ____________.
The molar conductivities at infinite dilution for sodium acetate, HCI and NaCl are 91 S cm2 mol-1, 425.9 S cm2 mol-1 and 126.4 S cm2 mol-1 respectively. The molar conductivity of acetic acid at infinite dilution is ______.
The resistance of a conductivity cell containing 0.001 M KCl solution at 298 K is 1500 Ω. What is the cell constant if conductivity of 0.001 M KCI solution at 298 K is 0.146 × 10−3 S cm−1?
In 2 hours, a certain current liberates 0.504 g hydrogen. How many grams of copper can be deposited by the same current flowing for the same time in a CuSO4 solution?
(Molar mass of Cu = 63.5 g mol−1, H2 = 2.0 g mol−1)
The distance between electrodes of a conductivity cell is 0.98 cm and area of cross section is 1.96 cm2. What is the cell constant?
If the conductivity of 0.08 M KCl solution is 2 × 10-2 Ω-1, what is the molar conductivity of the solution?
A conductivity cell dipped in 0.5 M KCl gives a resistance of 250 ohms. If the conductivity of KC I solution is 6.68 × 10-3 S cm-1, what is the cell constant?
The value of [H3O+] in mol lit–1 of 0.001 M acetic acid solution (Ka = 1.8 × 10–5) is ______.
Specific conductance of 0.1 M sodium chloride solution is 1.06 × 10−2 ohm−1 cm−1. Its molar conductance in ohm−1 cm2 mol−1 is ______.
What is the SI unit of conductivity?
Calculate molar conductivity for 0.5 M BaCl2 if its conductivity at 298K is 0.01 Ω–1 cm–1.
A conductivity cell filled with 0.02 M AgNO3 gives at 25°C resistance of 947 ohms. If the cell constant is 2.3 cm-1, what is the molar conductivity of 0.02 M AgNO3 at 25°C?
What is the SI unit of molar conductivity?
Explain the determination of molar conductivity of a weak electrolyte at infinite dilution or zero concentration using Kohlrausch's law.
Define cell constant. Give its units.
