Advertisements
Advertisements
प्रश्न
What is the coordination number in a square close packed structure in two dimensions?
पर्याय
2
3
4
6
उत्तर
4
Explanation:
Coordination number in a square closed packed structure in two dimensions is equal to 4 is shown as:
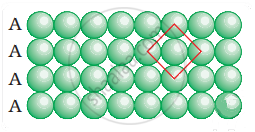
APPEARS IN
संबंधित प्रश्न
A cubic solid is made of two elements P and Q. Atoms of Q are at the corners of the cube and P at the body-centre. What is the formula of the compound? What are the coordination numbers of P and Q?
Ferric oxide crystallises in a hexagonal close-packed array of oxide ions with two out of every three octahedral holes occupied by ferric ions. Derive the formula of the ferric oxide.
In NaCl structure ____________.
Which of the following statement is not true about the hexagonal close packing?
Match the type of packing given in Column I with the items given in Column II.
| Column I | Column II |
| (i) Square close packing in two dimensions |
(a) Triangular voids |
| (ii) Hexagonal close packing in two dimensions |
(b) Pattern of spheres is repeated in every fourth layer |
| (iii) Hexagonal close packing in three dimensions |
(c) Coordination number 4 |
| (iv) Cubic close packing in three dimensions |
(d) Pattern of sphere is repeated in alternate layers |
With the help of a labelled diagram show that there are four octahedral voids per unit cell in a cubic close packed structure.
Total no. of voids in 0.5 mole of a compound forming hexagonal closed packed structure are:-
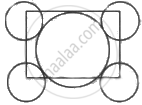
The packing efficiency of the two dimensional square unit cell shown below is:
A compound forms hexagonal close-packed structure. What is the total number of voids in 0.5 mol of it? How many of these are tetrahedral voids?
The number of tetrahedral and octahedral voids in a CCP array of 100 atoms are respectively:
