Advertisements
Advertisements
प्रश्न
Which of the following reactions of glucose can be explained only by its cyclic structure?
पर्याय
Glucose forms pentaacetate
Glucose reacts with hydroxylamine to form an oxime
Pentaacetate of glucose does not react with hydroxylamine
Glucose is oxidised by nitric acid to gluconic acid
उत्तर
Pentaacetate of glucose does not react with hydroxylamine
Explanation:
The pentaacetate of glucose does not react with hydroxylamine indicating the absence of free –CHO group. This property of glucose can be explained only by its cyclic structure.
APPEARS IN
संबंधित प्रश्न
What happens when glucose is treated with hydroxylamine?
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
Answer the following question.
What is the basic structural difference between glucose and fructose?
The spatial arrangement of the given molecule is denoted by:

Which of the following statements is incorrect regarding glucose?
Which one of the following compounds is different from the rest?
Which of the following properties of glucose cannot be explained by its open chain structure?
(i) Glucose does not form hydrogen sulphite with NaHSO3.
(ii) On oxidation with HNO3 glucose gives saccharic acid.
(iii) Glucose is found to exist in two different crystalline forms which are named as α and β.
Glucose is found to exist in two different α and β crystalline forms. These forms can be obtained by:
(i) The α form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(ii) The β form of glucose is obtained by crystallisation from a concentrated solution of glucose at 303 K.
(iii) The β form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
(iv) The α form is obtained by crystallisation from hot and saturated aqueous solution at 371 K.
Why does compound (A) given below not form an oxime?
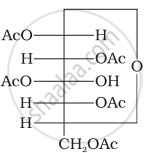
(A)
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
