Advertisements
Advertisements
प्रश्न
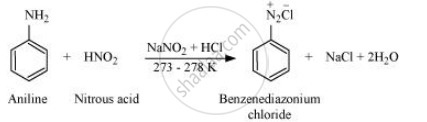
Write the reaction of aromatic primary amine with nitrous acid.
उत्तर
Aromatic amine react with nitrous acid (prepared in situ from NaNO2 and a mineral acid such as HCl) at 273 − 278 K to form stable aromatic diazonium salts, i.e., NaCl and H2O.
APPEARS IN
संबंधित प्रश्न
Accomplish the following conversion:
Benzyl chloride to 2-phenylethanamine
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Mention 'two' uses of propan-2-one.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Write structures of compounds A and B in each of the following reactions: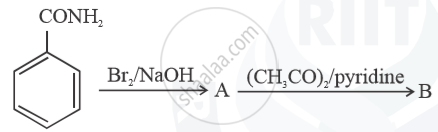
Answer the following
Write a reaction to convert acetic acid into methylamine.
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Which of the following does NOT give carbylamine test?
The source of nitrogen in Gabriel synthesis of amines is ______.
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Which of the following reactions are correct?
(i)
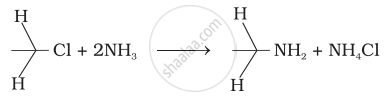
(ii)
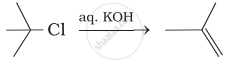
(iii)
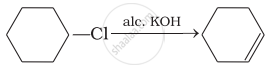
(iv)
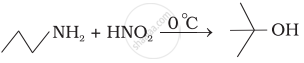
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
Suggest a route by which the following conversion can be accomplished.
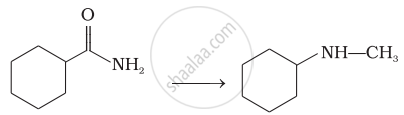
Describe Gabriel's phthalimide synthesis. (Give reaction)
Which of the following compound is expected to be most basic?
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write short note on the following:
Ammonolysis
