Advertisements
Advertisements
Question
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Solution
Van der Waals forces between closely packed linear polymeric chains are responsible for high-density polymer.
APPEARS IN
RELATED QUESTIONS
Explain the following term: Homopolymers
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Write the structures of the monomers used for getting the following polymers
Melamine – formaldehyde polymer
Answer the following in one sentence.
Identify 'B' in the following reaction:
\[\ce{H2N -(CH2)6 - NH2 + HOOC - (CH2)4 - COOH ->[N2][533 K]}\]'B'
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following.
Write the reaction of the formation of Terylene.
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Identify condensation polymers and addition polymers from the following.
\[\begin{array}{cc}\ce{-(CH2 - CH -)_{n}}\\
\phantom{....}|\\\ce{\phantom{.......}C6H5}
\end{array}\]
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Identify condensation polymers and addition polymers from the following.
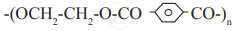
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Attempt the following:
What is meant by LDP and HDP? Mention the basic difference between the same with suitable examples.
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Nylon 6, 6 is a condensation polymer of hexamethylenediamine and _____________
Monomer of natural rubber is __________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Explain vulcanization of rubber.
Which of the following polymer is used in paints?
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
Which of the following is the monomer of neoprene?
Which of the following polymers is a heteropolymer?
Which among the following polymers can NOT be remoulded?
Identify the monomers used in the preparation of Novolac.
Which among the following monomers is used to prepare Teflon?
Which of the following polymers is prepared by using phenol?
Which among the following polymers is an example of addition polymer?
Which of the following pair of compounds is used as monomers for bakelite?
Identify addition polymer from the following.
Which among the following is an example of addition polymer?
The monomer of Teflon is ______.
F2C = CF2 is monomer of the polymer -
Which of the following polymer has ester linkage?
Which of the following is an example of polyester?
The catalyst used for the polymerisation of olefins is ______.
Identify the monomer used to prepare neoprene.
Which of the following is a polymer of enzyme?
Name and draw the structure of the repeating unit in natural rubber.
How the Bakelite is prepared? Give the steps involved in the preparation.
Name the compound which reacts with formaldehyde to produce ethyl alcohol.
Write the name of the monomer of natural rubber.
Another name of terylene is ______.
Name and draw structure of the repeating unit in natural rubber.
Write the structure and name of monomer of Nylon-6.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
