Advertisements
Advertisements
Question
Answer the following.
Write the reaction of the formation of Nylon 6.
Solution

APPEARS IN
RELATED QUESTIONS
Write any ‘two' uses of terylene.
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Write the monomers of the following polymer :
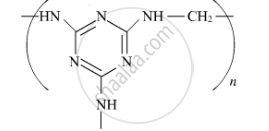
Choose the correct option from the given alternatives.
Which of the following is made up of polyamides?
Answer the following in one sentence.
Define the term: Vulcanization
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Answer the following.
Draw the structures of polymers formed from the following monomers
H2N–(CH2)5 – COOH
Identify condensation polymers and addition polymers from the following.
-(CO(CH2)4 - CONH(CH2)6NH-)n
Identify condensation polymers and addition polymers from the following.
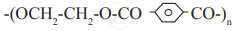
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Attempt the following:
Explain the vulcanisation of rubber. Which vulcanizing agents are used for the following synthetic rubber?
a. Neoprene
b. Buna-N
Write the reaction involved in the formation of:
Bakelite
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
write the structure of the monomer used in natural rubber.
Write chemical reaction for preparation of the following.
Buna-S
Write chemical reaction for preparation of the following.
Neoprene
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
Write chemical reactions for the preparation of high-density polythene.
The following structure represents the polymer:
\[\begin{array}{cc}
\ce{[-C-CH2-NH-C-(-CH2)5 NH -]_{{n}}}\\
\phantom{}||\phantom{.............}||\phantom{................}\\
\phantom{}\ce{O}\phantom{.............}\ce{O}\phantom{................}
\end{array}\]
Which among the following polymers is used for making handles of cooker?
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
Which of the following polymers is obtained from chloroprene?
Novolac is obtained from ____________.
Which of the following polymers is a heteropolymer?
Which of the following catalysts is used in preparation of terylene?
Which among the following catalysts is used in the preparation of dacron?
Which among the following monomers is used to prepare Teflon?
Which of the following polymers is prepared by using phenol?
Which of the following monomers is used in the manufacture of Neoprene rubber?
Which among the following is an example of addition polymer?
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Phenol and formaldehyde undergo condensation to give a polymar (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers?
F2C = CF2 is monomer of the polymer -
Which of the following polymer has ester linkage?
Which of the following is an example of polyester?
Which of the following polymers is synthesized using a free radical polymerisation technique?
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Identify the monomer used to prepare neoprene.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
How the Bakelite is prepared? Give the steps involved in the preparation.
Write the preparation of viscose rayon.
Write the structure of isoprene and the polymer obtained from it.
Another name of terylene is ______.
Name and draw structure of the repeating unit in natural rubber.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
Write the structure and name of monomer of Natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
