Advertisements
Advertisements
Question
Apply first law for an isothermal process.
Solution
For an isothermal process since the temperature is constant, the internal energy is also constant. This implies that dU or ∆U = 0.
For an isothermal process, the first law of thermodynamics can be written as follows,
Q = W
APPEARS IN
RELATED QUESTIONS
An ideal gas is taken through an isothermal process. If it does 2000 J of work on its environment, how much heat is added to it?
Draw a p-V diagram of the irreversible process.
Draw a p-V diagram showing negative work with varying pressure.
State the assumptions made for thermodynamic processes.
Apply first law for an adiabatic process.
Draw the PV diagram for the adiabatic process.
What is a cyclic process?
Can the given heat energy be completely converted to work in a cyclic process? If not, when can the heat can completely converted to work?
A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______
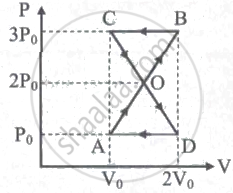
Among the amount of heat absorbed and the amount of work done by a system, ______
