Advertisements
Advertisements
Question
Complete the synthesis by giving missing starting material, reagent or product.
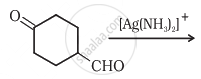
Solution
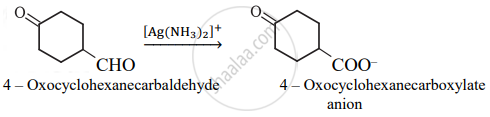
APPEARS IN
RELATED QUESTIONS
Predict the products of the following reactions :
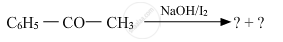
Give a simple chemical test to distinguish between the following pair of compounds :
CH3CH2CHO and CH3CH2COCH3
Give a simple chemical test to distinguish between the following pair of compounds:
Acetophenone and Benzophenone
Give a simple chemical test to distinguish between the following pair of compounds:
Phenol and Benzoic acid
Give a simple chemical test to distinguish between the following pair of compounds:
Benzoic acid and Ethyl benzoate
An organic compound contains 69.77% carbon, 11.63% hydrogen and rest oxygen. The molecular mass of the compound is 86. It does not reduce Tollens’ reagent but forms an addition compound with sodium hydrogensulphite and give positive iodoform test. On vigorous oxidation it gives ethanoic and propanoic acid. Write the possible structure of the compound.
Alkenes decolourise bromine water in presence of CCl4 due to formation of ______.
Which of the following compounds will give butanone on oxidation with alkaline \[\ce{KMnO4}\] solution?
Which of the following compounds gives a positive Tollen's test but negative Fehling's test?
Which sugar does not reduce Fehling's solution?
Fehilng's test is positive for
Ammonical silver nitrate solution is called
Which of the following tests/reactions is given by aldehydes as well as ketones?
A hydrocarbon (A) with molecular formula C5H10 on ozonolysis gives two products (B) and (C). Both (B) and (C) give a yellow precipitate when heated with iodine in presence of NaOH while only (B) give a silver mirror on reaction with Tollen’s reagent.
- Identify (A), (B) and (C).
- Write the reaction of B with Tollen’s reagent.
- Write the equation for iodoform test for C.
- Write down the equation for aldol condensation reaction of B and C.
Write chemical test to distinguish between the following compounds:
Phenol and Benzoic acid
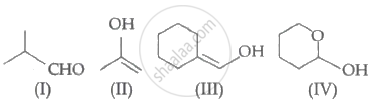
Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
The correct set of products obtained in the following reactions:
- \[\ce{RCN ->[reduction]}\]
- \[\ce{RCN ->[(i) CH3MgBr][(ii) H2O]}\]
- \[\ce{RNC ->[hydrolysis]}\]
- \[\ce{RNH2 ->[HNO2]}\]
Choose the reaction which is not possible:
