Advertisements
Advertisements
Question
Give a simple chemical test to distinguish between the following pair of compounds:
Phenol and Benzoic acid
Solution 1
Phenol and benzoic acid can be distinguished by ferric chloride test. In the ferric chloride test, phenol reacts with neutral FeCl3 to make an iron-phenol complex that turns violet.
\[\ce{\underset{Phenol}{6C6H5OH} + FeCl3 -> \underset{(Violet colour)}{\underset{Iron-phenol complex}{[Fe(OC6H5)6]^3-}} + 3H+ + 3Cl-}\]
But benzoic acid reacts with neutral FeCl3 to give a buff coloured ppt. of ferric benzoate.
\[\ce{\underset{Benzoic acid}{3C6H5OH} + FeCl3 ->\underset{(Buff coloured ppt)}{\underset{Ferric benzoate}{(C6H5COO)3Fe}} + 3HCl}\]
Solution 2
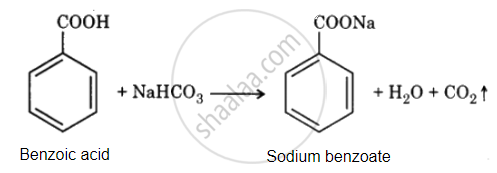
Benzoic acid reacts with NaHCO3 to release CO2 gas with effervescence, whereas phenol does not.
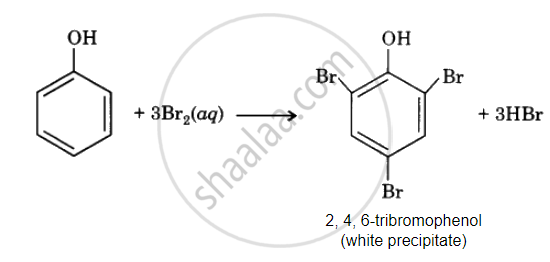
Phenol Br2 decolourises water to give white precipitate but does not give benzoic acid.
APPEARS IN
RELATED QUESTIONS
Give a simple chemical test to distinguish between the following pair of compounds:
Ethanal and Propanal
Distinguish between:
C6H5-COCH3 and C6H5-CHO
A and B are two functional isomers of compound C3H6O.On heating with NaOH and I2, isomer B forms yellow precipitate of iodoform whereas isomer A does not form any precipitate. Write the formulae of A and B.
Predict the products of the following reactions :
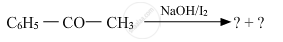
Propanal and Propanone
Out of CH3CH2 – CO – CH2 – CH3 and CH3CH2 – CH2 – CO – CH3, which gives iodoform test?
Give a simple chemical test to distinguish between the following pair of compounds:
Pentan-2-one and Pentan-3-one
Which of the following compounds will give butanone on oxidation with alkaline \[\ce{KMnO4}\] solution?
Which sugar does not reduce Fehling's solution?
Fehilng's test is positive for
Ammonical silver nitrate solution is called
Write chemical test to distinguish between the following compounds:
Phenol and Benzoic acid
The major products formed in the following reaction sequence A and B are:
In Tollen's test for aldehyde, the overall number of electrons(s) transferred to the Tollen's reagent formula \[\ce{[Ag(NH3)2]+}\] per aldehyde group to form silver mirror is ______. (Round off to the nearest integer)
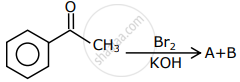
The reagent that can be used to distinguish acetophenone and benzophenone is ______.
An organic compound 'A' with the molecular formula C4H8O2 undergoes acid hydrolysis to form two compounds 'B' and 'C'. Oxidation of 'C' with acidified potassium permanganate also produces 'B'. Sodium salt of 'B' on heating with soda lime gives methane.
- Identify 'A', 'B' and 'C'.
- Out of 'B' and 'C', which will have higher boiling point? Give reason.
