Advertisements
Advertisements
Question
Convert bromoethane to propanamine.
Solution
\[\ce{\underset{Bromoethane}{CH3-CH2-Br} ->[alc. KCN] \underset{Propanenitrile}{CH3CH2CN} ->[LiAlH4] \underset{Propanamine}{CH3CH2CH2NH2}}\]
APPEARS IN
RELATED QUESTIONS
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
Arrange the compounds of the following set in order of reactivity towards SN2 displacement:
1-Bromobutane, 1-Bromo-2, 2-dimethylpropane, 1-Bromo-2-methylbutane, 1-Bromo-3-methylbutane
SN2 mechanism proceeds through intervention of ____________.
Which of the following undergoes nucleophilic substitution exclusively by SN1 mechanism?
Which of the following compound will undergo racemisation when reacts with aq. KOH?
(i)
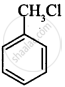
(ii)
CH3CH2CH2Cl
(iii)
\[\begin{array}{cc}
\ce{CH3}\phantom{..}\\
|\phantom{....}\\
\ce{CH3-CH-CH2Cl}
\end{array}\]
(iv)
\[\begin{array}{cc}
\phantom{..}\ce{H}\\
\phantom{..}|\\
\ce{CH3-C-Cl}\\
\phantom{..}|\\
\phantom{.....}\ce{C2H5}
\end{array}\]
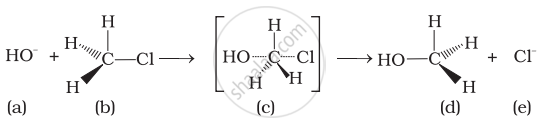
Which of the statements are correct about above reaction?
(i) (a) and (e) both are nucleophiles.
(ii) In (c) carbon atom is sp3 hybridised.
(iii) In (c) carbon atom is sp2 hybridised.
(iv) (a) and (e) both are electrophiles.
Cyanide ion acts as an ambident nucleophile. From which end it acts as a stronger nucleophile in aqueous medium? Give reason for your answer.
CCl4 is insoluble in water because:-
The decreasing order of reactivity of the following compounds towards nucleophilic substitution (SN2) is ______.
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]