Advertisements
Advertisements
Question
Derive a relation between ΔH and ΔU for a chemical reaction. Draw neat labelled diagram of calomel electrode. Resistance and conductivity of a cell containing 0.001 M KCI solution at 298K are 1500Ω and 1.46x10-4 S.cm-1 respectively.
Solution
Relation between ΔH and ΔU
The heat of reaction is given by enthalpy change
ΔH=H2 - H1
by defination H= U+PV
H1=U1+P1V1
H2=U2+P2V2
ΔH = (U1+P1V1) - (U2+P2V2)
=(U2-U1)+(P2V2-P1V1)
=ΔU+(P2V2-P1V1)(∵ΔU=U2-U1)
since PV=nRT
for initial state P1V1=n1RT
for final state P2V2=n2RT
P2V2-P1V1=n2RT-n1RT
=(n2-n)RT
=ΔnRT
where Δn = number of moles of gaseous product - number of moles of gaseous reactant.
| ΔH=ΔU+ΔnRT |
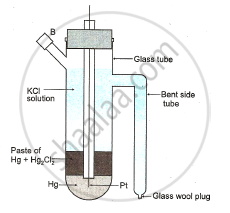
Diagram : Standard calomel electrode
Data: R 1500Ω
C=0.001M
k= 1.46x10-4Scm-1
To find: b = ?
Solution : `k=b/R`
b=kxR
=1.46x10-4x1500
b=2190x10-4 cm-1
| b = 0.219cm-1 |
APPEARS IN
RELATED QUESTIONS
Can you store copper sulphate solutions in a zinc pot?
Arrange the following metals in the order in which they displace each other from the solution of their salts.
\[\ce{Al, Cu, Fe, Mg}\] and \[\ce{Zn}\]
Construct a labelled diagram for the following cell:
`Zn|Zn^(2+)(1M)||H^+(1M)|H_(2(g,1atm))|Pt`
Why cannot we store AgNO3 solution in copper vessel?
How many moles of electrons are required for reduction of 2 moles of Zn2+ to Zn?
For the electrochemical cell:
M | M+ || X− | X,
E0(M+ | M) = 0.44 V, E0(X | X−) = 0.33 V
Which of the following is TRUE for this data?
If 'I' stands for the distance between the electrodes and 'a' stands for the area of cross-section of the electrode, `"l"/"a"` refers to ____________.
What is the SI unit tor electrochemical equivalent?
A solution of Cu(NO3)2 is electrolyzed between platinum electrodes using 0.1 Faraday electricity. How many moles of Cu will be deposited at the cathode?
Assertion: pure iron when heated in dry air is converted with a layer of rust.
Reason: Rust has the compositionFe3O4.
In the electrochemical cell: Zn|ZnSO4 (0.01 M)||CuSO4 (1.0 M)|Cu, the emf of this Daniel cell is E1. When the concentration of ZnSO4 is changed to 1.0 M and that CuSO4 changed to 0.01 M, the emf changes to E2. From the above, which one is the relationship between E1 and E2?
Cell equation: \[\ce{A + 2B^- -> A^{2+} + 2B}\]
\[\ce{A^{2+} + 2e^- -> A}\] E0 = +0.34 V and log10 k = 15.6 at 300 K for cell reactions find E0 for \[\ce{B^+ + e^- -> B}\]
Describe the electrolysis of molten NaCl using inert electrodes.
Describe the construction of Daniel cell. Write the cell reaction.
Can Fe3+ oxidises bromide to bromine under standard conditions?
Given: \[\ce{E^0_{{Fe^{3+}|Fe^{2+}}}}\] = 0.771 V
\[\ce{E^0_{{Br_{2}|Br^-}}}\] = −1.09 V
Is it possible to store copper sulphate in an iron vessel for a long time?
Given: \[\ce{E^0_{{Cu^{2+}|{Cu}}}}\] = 0.34 V and \[\ce{E^0_{{Fe^{2+}|{Fe}}}}\] = −0.44 V
Two metals M1 and M2 have reduction potential values of −xV and +yV respectively. Which will liberate H2 and H2SO4.
Reduction potential of two metals M1 and M2 are \[\ce{E^0_{{M_1^{2+}|M_1}}}\] = −2.3 V and \[\ce{E^0_{{M_2^{2+}|M_2}}}\] = 0.2 V. Predict which one is better for coating the surface of iron.
Given: \[\ce{E^0_{{Fe^{2+}|Fe}}}\] = −0.44 V
A copper electrode is dipped in 0.1 M copper sulphate solution at 25°C. Calculate the electrode potential of copper.
[Given: \[\ce{E^0_{{Cu^{2+}|Cu}}}\] = 0.34 V]
For the cell \[\ce{Mg_{(s)}|Mg^{2+}_{( aq)}||Ag^+_{( aq)}|Ag_{(s)}}\], calculate the equilibrium constant at 25°C and maximum work that can be obtained during operation of cell.
Given: \[\ce{E^0_{{Mg^{2+}|Mg}}}\] = −2.37 V and \[\ce{E^0_{{Ag^{+}|Ag}}}\] = 0.80 V
Electrode potential for Mg electrode varies according to the equation
`E_(Mg^(2+) | Mg) = E_(Mg^(2+) | Mg)^Θ - 0.059/2 log 1/([Mg^(2+)])`. The graph of `E_(Mg^(2+) | Mg)` vs `log [Mg^(2+)]` is ______.
Which of the following statement is correct?
An electrochemical cell can behave like an electrolytic cell when ______.
`E_(cell)^Θ` for some half cell reactions are given below. On the basis of these mark the correct answer.
(a) \[\ce{H^{+} (aq) + e^{-} -> 1/2 H_2 (g); E^Θ_{cell} = 0.00V}\]
(b) \[\ce{2H2O (1) -> O2 (g) + 4H^{+} (aq) + 4e^{-}; E^Θ_{cell} = 1.23V}\]
(c) \[\ce{2SO^{2-}_{4} (aq) -> S2O^{2-}_{8} (aq) + 2e^{-}; E^Θ_{cell} = 1.96V}\]
(i) In dilute sulphuric acid solution, hydrogen will be reduced at cathode.
(ii) In concentrated sulphuric acid solution, water will be oxidised at anode.
(iii) In dilute sulphuric acid solution, water will be oxidised at anode.
(iv) In dilute sulphuric acid solution, \[\ce{SO4^{2-}}\] ion will be oxidised to tetrathionate ion at anode.
What is electrode potential?
Consider the following diagram in which an electrochemical cell is coupled to an electrolytic cell. What will be the polarity of electrodes ‘A’ and ‘B’ in the electrolytic cell?
A galvanic cell has electrical potential of 1.1V. If an opposing potential of 1.1V is applied to this cell, what will happen to the cell reaction and current flowing through the cell?
Match the terms given in Column I with the units given in Column II.
| Column I | Column II |
| (i) Λm | (a) S cm-¹ |
| (ii) ECell | (b) m-¹ |
| (iii) K | (c) S cm2 mol-¹ |
| (iv) G* | (d) V |
The electrochemical cell stops working after some time because
Calculate the standard EMF ofa cell which involves the following cell reactions
\[\ce{Zn + 2 Ag+ -> Zn^{2+} + 2 Ag}\]
Given that \[\ce{E^{o}_{Zn/Zn^{2+}}}\] = 0.76 volt and \[\ce{E^{o}_{Ag/Ag^{+}}}\] = – 0.80 volt.
A current of 2.0 ampere passed for 5 hour through a molten salt deposits 22 g of the metal (Atomic mass = 177). The oxidation state of the metal in the metal salt is
Given the data at 25°C
Ag + I– → Agl + e–; E° = – 0.152 V
Ag → Ag+ + e–; E° = – 0.800 V
The value of log Ksp for Ag I is :-
The correct order of the mobility of the alkali metal ions. In aqueous solultion is
If the value of Ksp for Hg2Cl2 (s) is X then the value of X will be ____ where pX = - log X.
Given:
\[\ce{Hg2Cl2 + 2e- -> 2Hg(l) + 2Cl-}\], E° = 0.27 V
\[\ce{Hg+2 + 2e- -> 2Hg(l)}\] E° = 0.81 V
Which of the following is incorrect?
The number of moles of electrons passed when the current of 2 A is passed through a solution of electrolyte for 20 minutes is ______.
The two half cell reaction of an electrochemical cell is given as
\[\ce{Ag+ + e- -> Ag}\], `"E"_("Ag"^+//"Ag")^circ` = - 0.3995 V
\[\ce{Fe^{2+} -> Fe^{3+} + e-}\], `"E"_("Fe"^{3+}//"Fe")^{2+}` = - 0.7120 V
The value of EMF will be ______.
Galvanic cell is a device in which ______.
The cell constant of a conductivity cell is 0.146 cm-1. What is the conductivity of 0.01 M solution of an electrolyte at 298 K, if the resistance of the cell is 1000 ohm?
Why is anode in galvanic cell considered to be negative and cathode positive?
What is an electrochemical cell? What does it consist of?
What are electrochemical reactions?
State the term for the following:
Two metal plates or wires through which the current enters and leaves the electrolytic cell.
