Advertisements
Advertisements
Question
Describe the following:
Cross aldol condensation
Solution
When aldol condensation is carried out between two different aldehydes and/or ketones, it is called cross aldol condensation. If both of them contain a-hydrogen atoms, it gives a mixture of four products. This is illustrated below by the aldol reaction of a mixture of ethanal and propanal.
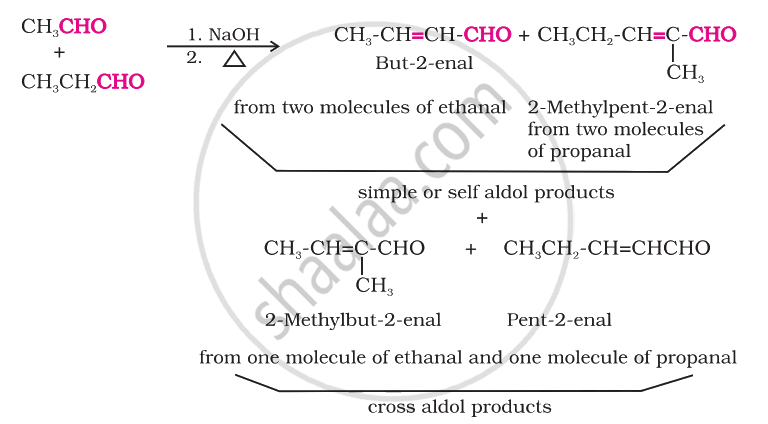
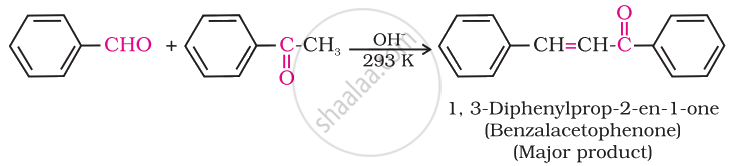
Ketones can also be used as one component in the cross-aldol reactions.
APPEARS IN
RELATED QUESTIONS
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
How will you bring about the following conversion?
Ethanal to but-2-enal
How will you convert ethanal into the following compound?
But-2-enal
How will you bring about the following conversion in not more than two steps?
Benzaldehyde to 3-Phenylpropan-1-ol
Complete the synthesis by giving missing starting material, reagent or product.
\[\begin{array}{cc}
\ce{C6H5CHO}\phantom{............}\\
\phantom{........}\ce{+\phantom{......}\ce{->[dil.NaOH][\Delta]}}\phantom{...}\\
\ce{CH3CH2CHO}\phantom{............}
\end{array}\]
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
What is substituted imine called?
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 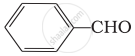
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
What product will be formed on reaction of propanal with 2-methylpropanal in the presence of \[\ce{NaOH}\]? What products will be formed? Write the name of the reaction also.
Cross aldol condensation occurs between
Which of the following gives aldol con~ensation reaction?
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
What is aldol condensation? Explain it with suitable examples.
