Advertisements
Advertisements
Question
Ethylbenzene is generally prepared by acetylation of benzene followed by reduction and not by direct alkylation. Think of a possible reason.
Solution
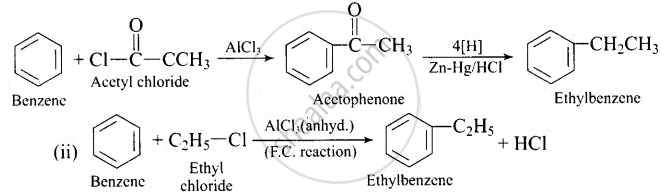
In (ii) reaction, it readily undergoes further alkylation to produce polysubs- tituted derivative.
APPEARS IN
RELATED QUESTIONS
Write the structure of the product of the following reaction:
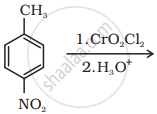
How will you prepare the given compound from benzene? You may use any inorganic reagent and any organic reagent having not more than one carbon atom.
p-Nitrobenzaldehyde
Write the reaction involved in the Stephen reduction
The oxidation of toluene to benzoic acid can be done using which of the following reagents.
What is the name of the given reaction of preparation of aldehyde?
\[\ce{C3COCl ->[H2][Pd/BaSO4] CH3CHO + HCl}\]
In the chromyl chloride test, the final step results in the formation of a yellow precipitate of the following:
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CH3}\\
\phantom{.....}|\\
\phantom{......}\ce{Cl}
\end{array}\] obtained by chlorination of n-butane, will be
The oxidation of toluene to benzaldehyde by chromyl chloride is called ______.
Explain the following reactions:
Stephan reaction
An organic compound with molecular formula \[\ce{C7H7NO2}\] exists in three isomeric forms, the isomer ‘A’ has the highest melting point of the three. ‘A’ on reduction gives compound ‘B’ with molecular formula \[\ce{C7H9N}\]. ‘B’ on treatment with \[\ce{NaNO2/HCl}\] at 0-5° C to form compound ‘C’. On treating C with \[\ce{H3PO2}\], it gets converted to D with formula \[\ce{C7H8}\], which on further reaction with \[\ce{CrO2Cl2}\] followed by hydrolysis forms ‘E’ \[\ce{C7H6O}\]. Write the structure of compounds A to E. Write the chemical equations involved.
