Advertisements
Advertisements
Question
Explain the factors affecting SN1 and SN2 mechanism.
Solution
Factors that affect the SN1 and SN2 mechanisms:
- Nature of substrate
- Nucleophilicity of the reagent
- Solvent polarity
i. Nature of substrate:
- SN2 mechanism: The transition state (T.S.) of the SN2 mechanism is pentacoordinate and thus crowded. As a result, SN2 mechanism is favoured in primary halides and least favoured in tertiary halides. Reactivity order of alkyl halides towards SN2 mechanism: 3° < 2° < 1°< CH3X.
- SN1 mechanism: A planar carbocation intermediate is formed in the SN1 reaction. It has no steric crowding as bulky alkyl groups can be easily accommodated in planar carbocation.
Formation of planar carbocation intermediate results in a relief from steric crowding present in the tertiary halide substrate.
Secondly, the carbocation intermediate is stabilized by +I effect of alkyl substituents and by the hyperconjugation effect of alkyl substituents containing α-hydrogens.
As a result, SN1 mechanism is most favoured in tertiary halides and least favoured in primary halides.
Reactivity order of alkyl halides towards SN1 mechanism: 3° > 2° > 1°> CH3X.
ii. Nucleophilicity of the reagent:
- SN2 mechanism: A more powerful nucleophile attacks the substrate faster and favours the SN2 mechanism.
- SN1 mechanism: The rate of the SN1 mechanism is independent of the nature of nucleophile as nucleophile does not react in the slow step of SN1. It waits till the carbocation intermediate is formed, and reacts fast with it.
iii. Solvent polarity:
- SN2 mechanism: Polar protic solvents usually decrease the rate of an SN2 reaction. In the rate-determining step of the SN2 mechanism, the substrate, as well as nucleophile, is involved.
A polar solvent stabilizes nucleophile (one of the reactants) by solvation. Thus, solvent deactivates the nucleophile by stabilizing it. Hence, aprotic solvents or solvents of low polarity will favour the SN2 mechanism. - SN1 mechanism: SN1 mechanism proceeds via the formation of a carbocation intermediate. A good ionizing solvent, polar solvent, stabilizes the ions by solvation.
Solvation of carbocation is relatively poor and solvation of anion is particularly important. Anions are solvated by hydrogen-bonding solvents, that is, protic solvents.
Thus, the SN1 reaction proceeds more rapidly in polar protic solvents than in aprotic solvents.
APPEARS IN
RELATED QUESTIONS
from the following pair would undergo SN2 faster from the other?
a. CH3CH2CH2I b. CH3CH2CH2Cl
Complete the following reaction giving major products.
\[\ce{CH3 - CH = CH2 ->[HBr][peroxide] A ->[alc. KOH] B}\]
Convert the following:
Benzyl alcohol to benzyl cyanide
Convert the following:
Ethanol to propane nitrile
Convert the following:
2-Chloropropane to propan-1-ol
HCl is added to a hydrocarbon ‘A’ \[\ce{(C4H8)}\] to give a compound ‘B’ which on hydrolysis with aqueous alkali forms tertiary alcohol ‘C’ \[\ce{(C4H10O)}\]. Identify ‘A’ ,‘B’ and ‘C’.
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{2-Bromobutane->[Alc.KOH]A->[][Br2]B->[][NaNH2]C}\]
Complete the following reaction sequence by writing the structural formulae of the organic compound 'A', 'B' and 'C'.
\[\ce{Isopropyl alcohol ->[\triangle][PBr3] A ->[][NH3 excess] B}\]
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
Propane nitrile can be prepared by heating ____________
The following will react faster by SN1 mechanism
Major product of the following reaction is _______________
\[\ce{CH3-CH2-Mg-Br + NH3 ->?}\]
Nucleophilic substitution reaction of 2, 4-dinitrochlorobenzene is faster than p-nitrochlorobenzene. Give reason.
Write the correct order of increasing ease of dehydrohalogenation.
\[\ce{\underset{\text{(I)}}{CH3 - CH2 - CH2 - Cl}}\]
\[\ce{\underset{\text{(II)}}{CH3 - CH(Cl) - CH3}}\]
\[\begin{array}{cc}
\ce{CH3}\\
|\\
\ce{CH3-C-Cl}\phantom{...}\\
|\\
\ce{CH3}\\
\ce{(III)}
\end{array}\]
Explain aqueous alkaline hydrolysis of tert. butyl bromide.
Which of the following is a primary halide?
Ethyl bromide undergoes the following reaction:
\[\ce{\underset{Ethyl bromide}{C2H5Br} + \underset{(aq.)}{KOH} ->[\Delta] \underset{Ethyl alcohol}{C2H5OH} + KBr}\]
Which of the following is a WRONG statement?
Which of the following is least reactive towards nucleophilic addition?
Dehydrohalogenation of an alkyl halide is ____________.
Nitroalkanes are obtained in laboratory from primary or secondary alkyl halides by the action of ______.
What type of hybridisation is present in carbocation formed during the alkaline hydrolysis of 1- bromo-1- phenyl ethane?
Which of the following is FALSE regarding SN2 reaction mechanism?
Observe the following and answer the questions given below:
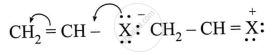
Name the type of halogen derivative.
What is the action of following on ethyl bromide?
alcoholic sodium hydroxide
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether] A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
