Advertisements
Advertisements
Question
Explain the inverse linear dependence of stopping potential on the incident wavelength in a photoelectric effect experiment.
Solution
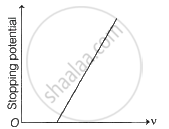
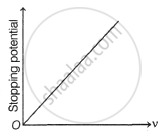
Hence, it follows that as `1/lambda` increases, V0 increases.
The plot of V0 verses `1/lambda` is linear. This is due to the fact that the energy associated with a quantum of radiation (photon) is directly proportional to its frequency and thus inversely proportional to its wavelength of radiation.
APPEARS IN
RELATED QUESTIONS
What is the photoelectric effect?
Using the values of work function given in the following table, tell which metal will require the highest frequency of incident radiation to generate photocurrent.
Typical values of work function for some common metals
| Metal | Work function (in eV) |
| Potassium | 2.3 |
| Sodium | 2.4 |
| Calcium | 2.9 |
| Zinc | 3.6 |
| Silver | 4.3 |
| Aluminium | 4.3 |
| Tungsten | 4.5 |
| Copper | 4.7 |
| Nickel | 5.0 |
| Gold | 5.1 |
The threshold wavelength of tungsten is 2.76 x 10-5 cm.
(a) Explain why no photoelectrons are emitted when the wavelength is more than 2.76 x 10-5 cm.
(b) What will be the maximum kinetic energy of electrons ejected in each of the following cases
(i) if ultraviolet radiation of wavelength λ = 1.80 × 10-5 cm and
(ii) radiation of frequency 4 x 1015 Hz is made incident on the tungsten surface?
As the intensity of incident light increases ______
The minimum frequency for photoelectric effect on metal is 7 × 1014 Hz, Find the work function of the metal.
The energy of a photon is 2 eV. Find its frequency and wavelength.
When a photon enters glass from air, which one of the following quantity does not change?
For photoelectric emission from certain metal, the cut-off frequency is v. If radiation of frequency 2v impinges on the metal plate, the maximum possible velocity of the emitted electron will be (m is the electron mass) ____________.
When a light of wavelength 4000 Å falls on a photoelectric emitter, photoelectrons are liberated. For another emitter, light of wavelength 6000 Å is sufficient for photo emission. The work functions of the two emitters are in the ratio of ____________.
Threshold frequency for a metal is 1015 Hz. Light of `lambda` = 4000 Å falls on its surface. Which of the following statements is correct?
The work function of a metal is 1.6 x 10-19 J. When the metal surface is illuminated by the light of wavelength 6400 Å, then the maximum kinetic energy of emitted photo-electrons will be (Planck's constant h = 6.4 x 10-34 Js) ____________.
Threshold wavelength for lithium metal is 6250 Å. For photoemission, the wavelength of the incident light must be ______.
When certain metal surface is illuminated with a light of wavelength A., the stopping potential is V, When the same surface is illuminated by light of wavelength 2λ, the stopping potential is `("V"/3)`. The threshold wavelength for the surface is ______.
In photoelectric effect, graph of saturation current versus frequency of light is plotted. The nature of the graph will be ____________.
The photon of frequency vis incident on a metal surface whose threshold frequency is v0. The kinetic energy of the emitted photoelectrons will be ______.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
A light of wavelength '`lambda`' and intensity 'I' falls on photosensitive material. If 'N' photoelectrons are emitted, each with kinetic energy E, then ____________.
When light of wavelength '`lambda`' is incident on photosensitive surface, photons of power 'P' are emitted. The number of photons (n) emitted in 't' second is (h = Planck's constant, c = velocity of light in vacuum) ____________.
When the work function of a metal increases, maximum kinetic energy of emitted photoelectrons ____________.
In a photoelectric experiment, ultraviolet light of wavelength 280 nm is used with a lithium cathode having work function Φ = 2.5 eV. If the wavelength of incident light is switched to 400 nm, find out the change in the stopping potential.
(h = 6.63 × 10-34 Js, c = 3 × 108 ms-1)
The radiation emitted, when an electron jumps from n = 3 to n = 2 orbit is a hydrogen atom, falls on a metal to produce photoelectron. The electrons from the metal surface with maximum kinetic energy are made to move perpendicular to a magnetic field of `1/320`T in a radius of 10-3m. Find the 320 work function of metal:
For a given photosensitive material and frequency (> threshold frequency) of incident radiation, the photoelectric current varies with the intensity of incident light as:
In a photocell, frequency of incident radiation is increased by keeping other factors constant (v > v0), the stopping potential ______.
The following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (ν) for a given metal. The correct variation is shown in graph [ν0 = threshold frequency].
|
(A) |
(B) |
|
(C) |
(D) |
Photoelectric emission is observed from a metallic surface for frequencies ν1 and ν2 of the incident light rays (ν1 > ν2). If the ratio of the maximum value of the kinetic energy of the photoelectrons emitted in the first case to that in the second case is 2 : K, then the threshold frequency of the metallic surface is ______.
When monochromatic light of frequency v1 falls on a metal surface, the stopping potential required is found to be V1. If the radiation of frequency v2 is incident on the surface, the stopping potential required V2 is ______. (v2 > v1)