Advertisements
Advertisements
Question
For a given ideal gas 6 × 105 J heat energy is supplied and the volume of gas is increased from 4 m3 to 6 m3 at atmospheric pressure. Calculate
- the work done by the gas
- change in internal energy of the gas
- graph this process in PV and TV diagram
Solution
Heat energy supplied to gas Q = 6 × 105 J
Change in volume ∆V = (6 – 4) = 2 m3
1 atm = 1.013 × 105 Nm−2
a. Work done by the gas W = P × ∆V
= 1.013 × 105 × 2
= 2.026 × 105
W = 202.6 kJ
b. Change in internal energy of the gas
∆U = Q − P. ∆V
= 6 × 105 − (202.6 × 103)
= 6 × 105 − 2.026 × 105
= 3.974 × 105
∆U = 397.4 kJ
c.
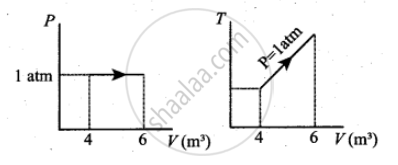
APPEARS IN
RELATED QUESTIONS
For work done to be reversible, the process should be ______
Explain the cyclic process.
An ideal gas of volume 2 L is adiabatically compressed to (1/10)th of its initial volume. Its initial pressure is 1.01 x 105 Pa, calculate the final pressure. (Given 𝛾 = 1.4)
Write a note on free expansion.
Give an expression for work done in an isothermal process.
Draw the PV diagram for the adiabatic process.
Explain in detail the isothermal process.
A thermodynamic system undergoes cyclic process ABCDA as shown in the figure. The work done by the system is ______
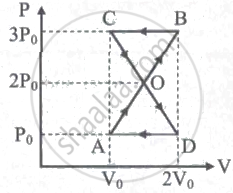
For an isothermal expansion of a perfect gas, the value of `(Delta "P")/"P"` is equal to ____________.
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
