Advertisements
Advertisements
Question
Give reason for the following:
Temperature remaining constant the product of the vol. & the press, of a given mass of dry gas is a constant.
Solution
According to Boyle's Law V α `1/"p"`
V = K.`1/"p"`
∴ P × V = K = a constant
∴ Product of volume and pressure of a given mass of dry gas is constant. [at constant temperature]
APPEARS IN
RELATED QUESTIONS
Estimate the average thermal energy of a helium atom at the temperature of 10 million Kelvin (the typical core temperature in the case of a star).
What do you understand by gas?
A gas occupies 500 cm3 at a normal temperature. At what temperature will the volume of the gas be reduced by 20% of its original volume, the pressure is constant?
Name or state the following:
The absolute temperature value corresponding to 35°C.
The average energy per molecule is proportional to ______
The equation of state for 2g of oxygen at a pressure 'P' and temperature 'T', when occupying a volume 'V' will be ______.
Estimate the average thermal energy of a helium atom at room temperature (27 °C).
Which of the following diagrams (Figure) depicts ideal gas behaviour?
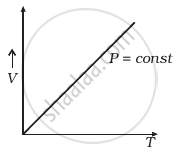 (a) |
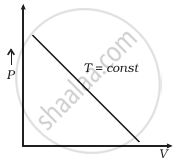 (b) |
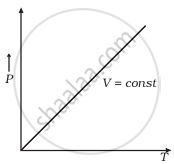 (c) |
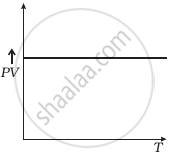 (d) |
At room temperature, a diatomic gas is found to have an r.m.s. speed of 1930 ms-1. The gas is ______.
For a wave, y = 0.0002 sin`[2pi(110"t"-x/3)+pi/3]` is travelling in a medium. The energy per unit volume being transferred by wave if density of medium is 1.5 kg/m3, is ______.
