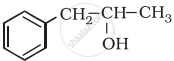Advertisements
Advertisements
Question
Identify allylic alcohols in the following examples.
Options
\[\begin{array}{cc}
\ce{CH3}\phantom{.}\\
|\phantom{....}\\
\ce{CH3 - C - CH2OH}\\
|\phantom{....}\\
\ce{CH3}\phantom{.}
\end{array}\]H2C = CH – CH2OH
CH3 – CH2 – CH2 – OH
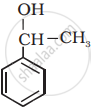
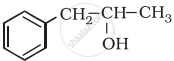
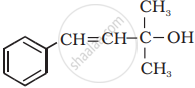
Solution
(ii) H2C = CH – CH2OH
(vi)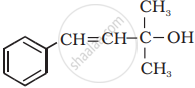
APPEARS IN
RELATED QUESTIONS
Give the structures and IUPAC names of monohydric phenols of molecular formula C7H8O.
The alcohol used in thermometers is _______.
(A) methanol
(B) ethanol
(C) propanol
(D) butanol
Why ethanol has the higher boiling point than ethane?
Write balanced chemical equation for the following :
Action of sodium metal on ethanol.
Which of the following is dihydric alcohol?
How many isomers of C5H11OH will be primary alcohols?
How many alcohol(s) with molecular formula \[\ce{C4H10O}\] is chiral in nature?
Number of metamers represented by molecular formula C4H10O is ____________.
Among the following compounds, the strongest acid is:
Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
\[\ce{n-Butane ->[AlCl3][HCl] [X] ->[KMnO4] [Y], [Y]}\] is
Glycerol is not used in which of the following cares
Main constituent of dynamite is.
Washing soap can be prepared by saponification with alkali of the oil.
Explain the classification of alcohol and phenol.
Classify the following as primary, secondary and tertiary alcohol:
H2C = CH – CH2OH
Classify the following as primary, secondary and tertiary alcohol:
CH3 – CH2 – CH2 – OH
Classify the following as primary, secondary and tertiary alcohol: