Advertisements
Advertisements
Question
Name the following:
The middle region of the blast furnace.
Solution
Zone of heat absorption
APPEARS IN
RELATED QUESTIONS
Answer the following questions with respect to the electrolytic process in the extraction of aluminum:
Identify the components of the electrolyte other than pure alumina and the role played by each
For the substance given below, describe the role played in the extraction of aluminium.
Cryolite
Give the chemical names and formulae of the main ores of zinc.
Give reason for the following:
Extraction of aluminium was very difficult in the beginning.
For the substance listed below, explain its role in the extraction of aluminium: Cryolite
For the substance listed below, explain its role in the extraction of aluminium: Graphite
Aluminium is extracted from its chief ore bauxite. The ore is first purified and then the metal is extracted from it by electrolytic reduction.
Name a chemicals used for dissolving aluminium oxide. In which state of sub-division is the chemical used?
Name the following :
A compound added to lower the fusion temperature of electrolytic bath in the extracton of aluminium.
Name the following:
A metal which is always present in the amalgam.
The following sketch illustrates the process of conversion of Alumina to Aluminium:
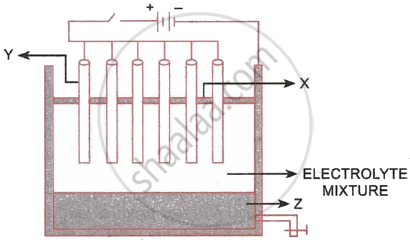
- Name the constituent of the electrolyte mixture which has a divalent metal in it.
- Name the powdered substances ‘X’ sprinkled on the surface of the electrolyte mixture.
- What is the name of the process?
- Write the reactions taking place at the electrodes ‘Y’ (anode) and ‘Z’ (cathode), respectively.
