Advertisements
Advertisements
Question
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.
Solution
Conc. H2SO4 or H3PO4 at 433-443 K is used as a reagent in the dehydration of propan-2-ol to propene.
APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Predict the major product of acid catalysed dehydration of butan-1-ol.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
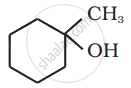
Show how you would synthesise the following alcohol from an appropriate alkene?
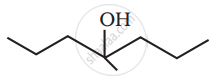
Show how you would synthesise the following alcohol from an appropriate alkene?
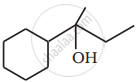
Write the structures of A, B and C in the following reactions :
Write the structure of main compounds A and B in the following reaction:
\[\ce{CH3CH2CN->[CH3MgBRH/3O+]A->[LiAIH4]B}\]
Identify ‘C’ in the following:
Magnesium wire continues to burn in the atmosphere of CO2 because
Select the acid(s) which cannot be prepared by Grignard reagent.
Alkaline hydrolysis of an alkyl halide can be preferably carried out using ______.
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
Write the mechanism of acid dehydration of ethanol to yield ethene.
