Advertisements
Advertisements
Question
How are polythene and neoprene prepared?
Solution
The antibiotic which is effective against a wide range of gram positive and gram negative bacteria known as broad spectrum antibiotic.
Preparating polyethene : The monomer used is ethene. It is an addition polymer. It is of two types.
\[\ce{n CH2 = CH2 ->[Δ] [-CH2 - CH2-]_n}\]
Preparating of Neoprne : The monomer used is chloroprene (2-chlorobuta-1,3-diene). It is superior to natural rubber and resistant to chemical action. It is used in the manufacture of chemical containers , conveyor belts, gaskets etc.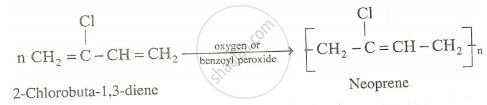
APPEARS IN
RELATED QUESTIONS
What is the role of t-butyl peroxide in the polymerization of ethene?
Write the mechanism of free radical polymerization of ethene.
Which of the following is a natural polymer ? Buna-S, Proteins, PVC
Write the free radical mechanism for the polymerisation of ethene.
Write chemical reaction to prepare the Dextron polymer:
Dextron
Which of the following statements is not true about low-density polythene?
What is the role of benzoyl peroxide in addition polymerisation of alkenes? Explain its mode of action with the help of an example.
Why should the monomers used in addition polymerisation through free radical pathway be very pure?
Match the polymer of column I with correct monomer of column II.
| Column I | Column II |
| (i) High density polythene | (a) Isoprene |
| (ii) Neoprene | (b) Tetrafluoroethene |
| (iii) Natural rubber | (c) Chloroprene |
| (iv) Teflon | (d) Acrylonitrile |
| (v) Acrilan | (e) Ethene |
Match the polymers given in Column I with the preferred mode of polymerisation followed by their monomers.
| Column I | Column II |
| (i) Nylon-6,6 | (a) Free radical polymerisation |
| (ii) PVC | (b) Ziegler-Natta polymerisation or coordination polymerisation |
| (iii) HDP | (c) Anionic polymerisation |
| (d) Condensation polymerisation |
Assertion: Olefinic monomers undergo addition polymerisation.
Reason: Polymerisation of vinyl chloride is initiated by peroxides/ persulphates.
Assertion: Polytetrafluoroethene is used in making non-stick cookwares.
Reason: Fluorine has highest electronegativity.
Low density polythene and high density polythene, both are polymers of ethene but there is marked difference in their properties. Explain.
By which reaction ethene is obtained from ethyne?
Which one of the following can be used as monomer in a polymerisation reaction?
The Ziegler Natta catalyst are
Which of the following is chain-growth polymer?
Which one of the following polymers are prepared by addition polymerisation?
Which one of the following polymers is prepared by addition polymerisation?
\[\ce{X + C + Cl2 ->[High temperature][of about 1000 K] Y + CO}\];
\[\ce{Y + 2H2O -> Z + 2HCl}\]
Compound Y is found in polymeric chain structure and is an electron-deficient Molecule. Y must be:
Which of the following statements about low density polythene is false?
