Advertisements
Advertisements
Question
Show the covalent bond formation in nitrogen molecule.
Solution
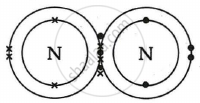
APPEARS IN
RELATED QUESTIONS
Explain why carbon forms compounds mainly by covalent bond.
Name the element whose one of the allotropic forms is buckminsterfullerene.
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many hexagons of carbon atoms are present in one molecule of buckminsterfullerene?
State one major difference between covalent and ionic bonds and give one example each of covalent and ionic compounds.
What type of bonds are present in H2 molecule? Draw their electron-dot structures.
Draw the electron-dot structure of HCl compound and state the type of bonding.
Describe the structure of graphite with the help of a labelled diagram.
Give the formulae of the chlorides of the elements X and Y having atomic numbers of 3 and 6 respectively. Will the properties of the two chlorides be similar or different? Explain your answer.
What happens when methane (natural gas) burns in air? Write the chemical equation of the reaction involved.
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Water
Give examples for the following:
Two gaseous non polar compounds.
Which of the following is a common characteristic of a covalent compound?
Draw the electron dot diagram and structure of nitrogen molecule.
Name two compounds that are covalent when taken pure but produce ions when dissolved in water.
What are Allotropes? Name any two allotropic forms of carbon. Give one use of it.
Number of valence electrons in a carbon atom is _______.
Give an example for each of the following statement.
A compound in which three covalent bonds are formed.
Which of the following is the purest form of carbon?
Although metals form basic oxides, which of the following metals form an amphoteric oxide?
A molecule of ammonia (NH3) has
