Advertisements
Advertisements
Question
State the observation for action of dilute hydrochloiric acid or iron (II) sulphate.
Solution
Dilute hydrochloric acid decomposes iron(II) sulphide to produce iron(II) chloride and hydrogen sulphide having rotten egg smell.
FeS + 2HCl → FeCl2 + H2S
APPEARS IN
RELATED QUESTIONS
Certain blank spaces are left in the following table and these are labelled as A, B, C, D
and E. Identify each of them
| Lab preparation of | Reactants used | Products formed | Drying Agent | Method of collection |
|
| 1 | HCl gas | NaCl + H2SO4 |  |
conc. H2SO4 | 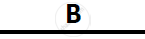 |
| 2 | NH3 gas |
|
Mg(OH)2 NH3 |
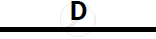 |
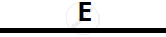 |
Identify the gas evolved and give the chemical test in the following cases
Dilute hydrochloric acid reacts with sodium sulphite.
Name the drying agents used in drying hydrogen chloride gas.
Name the drying agents phosphorus pentoxide and calcium oxide are good drying agent but they cannot be used to dry hydrogen chloride gas. Why?
Explain why when the stopper of a bottle full of hydrogen chloride gas is opened, there are fumes in the air.
Explain why a solution of hydrogen chloride in water turns blue litmus red and conducts electricity, while a solution of the same gas in toluene:
- has no effect on litmus, and
- does not conduct electricity.
The drying agent used to dry \[\ce{HCl}\] gas is ______.
Draw a labelled diagram and explain the laboratory preparation of hydrogen chioride gas.
What are the important precautions?
Explain, why (or give reasons for)
HCI gas does not conduct electricity, but hydrochloric acid conducts electricity.
What is aqua-regia?
Mention the reaction condition and give balanced equation to obtain: Cl2 gas from HCI gas.
Mention the reaction condition and give balanced equation to obtain : HCl gas from common salt
Write balanced equation for the reaction between dilute hydrochloric acid and sodium sulphite.
Write the equation for :
The preparation of hydrogen chloride from sodium chloride and sulphuric acid. State whether the sulphuric acid should be concentrated or dilute.
What property of hydrogen chloride is demonstrated when it is collected by downward delivery (upward displacement) ?
Hydrogen chloride gas, being highly soluble in water, is dried by ______.
One chemical test that would enable you to distinguish between the following pair of chemicals. Describe what happens with each chemical or state 'no visible reaction'.
Sodium sulphate solution and sodium chloride solution.
State a relevant reason for the following:
Hydrogen chloride gas cannot be dried over quick lime.
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
