Advertisements
Advertisements
Question
Give the formula and describe the structure of a noble gas species which is isostructural with:
`IBr_2^(-)`
Solution 1
XeF2 is isoelectronic to `IBr_2^(-)` and has a linear structure.
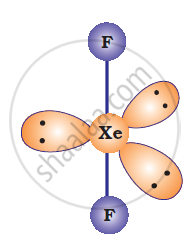
Solution 2
IBr2–: In IBr2–, central atom I has eight electrons. Two of these are utilized in forming two single bonds with two Br atom. Six remaining electrons constitutes three lone pairs. It is arranged in linear structure.

IBr2– has 22 valence electrons. A noble gas species having 22 valence electrons is XeF2 (8+2 x 7=22).
XeF2 is also linear.
APPEARS IN
RELATED QUESTIONS
Draw the structures of the following molecules: XeF4
What happens when: XeF4 reacts with SbF5?
Which noble gas is used in filling balloons for meteorological observations?
Write the structures of the following molecules: XeOF4
How are xenon fluorides XeF2, XeF4 and XeF6 obtained?
Give the formula and describe the structure of a noble gas species which is isostructural with:
`BrO_3^(-)`
Draw structures of the following:
XeF4
Draw the structure of XeF4.
Draw structure and name the shape of bromine trifluoride.
Explain the trend in the following atomic properties of group 16 elements:
Electron gain enthalpy
Which of the following reactions is an example of redox reaction?
Match the compounds given in Column I with the hybridisation and shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) XeF6 | (1) sp3d3 – distorted octahedral |
| (B) XeO3 | (2) sp3d2 – square planar |
| (C) XeOF4 | (3) sp3 – pyramidal |
| (D) XeF4 | (4) sp3d2 – square pyramidal |
The order of increasing sizes of atomic radii among the elements O, S, Se and As is:
Match List - I with List - II:
| List - I | List - II | ||
| (Species) | (Number of lone pairs of electrons on the central atom) |
||
| (A) | XeF2 | (i) | 0 |
| (B) | XeO2F2 | (ii) | 1 |
| (C) | XeO3F2 | (iii) | 2 |
| (D) | XeF4 | (iv) | 3 |
Choose the most appropriate answer from the options given below:
\[\ce{XeF6 + H2O ->[Partial][Hydrolysis] \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
\[\ce{XeF4 + H2O - \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
