Advertisements
Advertisements
Question
The conductivity of 0.20 mol L−1 solution of KCl is 2.48 × 10−2 S cm−1. Calculate its molar conductivity and degree of dissociation (α). Given λ0 (K+) = 73.5 S cm2 mol−1 and λ0 (C1−) = 76.5 S cm2 mol−1.
Solution
Conductivity of KCl solution = 2.48×10–2 S cm–1
Concentration of KCl solution = 0.20 mol L–1
= 0.20 × 1000 mol cm–3
= 200 mol cm–3
Molar conductivity
`Lambda_m=k/c`
`=(2.48xx10^(-2)" S "cm^(-1))/(200" mol "cm^(-3))`
`=124xx10^(-6)" S "cm^2" mol"^(-1)`
Given:
`lambda_(K^+)^@=73.5" S "cm^2" mol"^(-1)`
`lambda_(Cl^-)^@=76.5" S "cm^2" mol"^(-1)`
Degree of dissociation
`alpha=Lambda_m/Lambda_m^@" "" "" "" ".......("i")`
`Lambda_(m(KCl))^@=lambda_(K+)^@+lambda_(Cl^-)^@`
`=73.5+76.5`
`=150" S "cm^2" mol"^(-1)`
`"Substituting the values of "Lambda_m" and "Lambda_(m(KCl))^@" in (i), we get"`
`alpha=(124xx10^(-6)" S "cm^2" mol"^(-1))/(150" S "cm^2" mol"^(-1))=0.82xx10^(-6)`
RELATED QUESTIONS
The conductivity of 0.001 mol L-1 solution of CH3COOH is 3.905× 10-5 S cm-1. Calculate its molar conductivity and degree of dissociation (α) Given λ°(H+)= 349.6 S cm2 mol-1 and λ°(CH3COO)= 40.9S cm2mol-1.
Why does the conductivity of a solution decrease with dilution?
10.0 grams of caustic soda when dissolved in 250 cm3 of water, the resultant gram molarity of solution is _______.
(A) 0.25 M
(B) 0.5 M
(C) 1.0 M
(D) 0.1 M
Define the following terms :
Limiting molar conductivity
How can you determine limiting molar conductivity, 0 m for strong electrolyte and weak electrolyte?
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2), following curves are obtained for two electrolytes A and B:
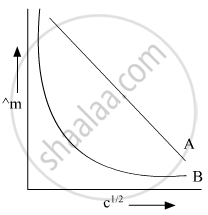
Answer the following:
(i) Predict the nature of electrolytes A and B.
(ii) What happens on extrapolation of ∧m to concentration approaching zero for electrolytes A and B?
\[\ce{Λ^0_m}_{(NH_4OH)}\] is equal to ______.
\[\ce{Λ^0_m H2O}\] is equal to:
(i) \[\ce{Λ^0_m_{(HCl)} + \ce{Λ^0_m_{(NaOH)} - \ce{Λ^0_m_{(NaCl)}}}}\]
(ii) \[\ce{Λ^0_m_{(HNO_3)} + \ce{Λ^0_m_{(NaNO_3)} - \ce{Λ^0_m_{(NaOH)}}}}\]
(iii) \[\ce{Λ^0_{(HNO_3)} + \ce{Λ^0_m_{(NaOH)} - \ce{Λ^0_m_{(NaNO_3)}}}}\]
(iv) \[\ce{Λ^0_m_{(NH_4OH)} + \ce{Λ^0_m_{(HCl)} - \ce{Λ^0_m_{(NH_4Cl)}}}}\]
Molar conductivity of ionic solution depends on:
(i) temperature.
(ii) distance between electrodes.
(iii) concentration of electrolytes in solution.
(iv) surface area of electrodes.
The solution of two electrolytes A and B are diluted. ^m of B increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Give a reason.
