Advertisements
Advertisements
Question
Calculate e.m.f. and ∆G for the following cell:
Mg (s) |Mg2+ (0.001M) || Cu2+ (0.0001M) | Cu (s)
`"Given :" E_((Mg^(2+)"/"Mg))^0=−2.37 V, E_((Cu^(2+)"/"Cu))^0=+0.34 V.`
Solution
For the given cell representation, the cell reaction will be
Mg(s) + Cu2+(0.0001 M) → Mg2+(0.001 M) + Cu(s)
The standard emf of the cell will be given by
= 0.34 - (-2.37)
= 2.71 V
The Nernst equation for the cell reaction at 25 ºC will be
`E_(Cell)=E_(Cell)^@-(0.059/n)"log"([Mg^(2+)])/([Cu^(2+)])`
`=2.71-0.059/2"log"0.001/0.0001`
`=2.71-0.02955(log10)`
`=2.71-0.02955(1)`
`=2.68045V~~2.68V`
We know
∆G = −nFEcell
=−2×96500×2.68
=−517240 J mol−1
=−517.24 kJ mol−1
RELATED QUESTIONS
Calculate Ecell and ΔG for the following at 28°C :
Mg(s) + Sn2+( 0.04M ) → Mg2+( 0.06M ) + Sn(s)
E°cell = 2.23V. Is the reaction spontaneous ?
Calculate E°cell for the following reaction at 298 K:
2Al(s) + 3Cu+2(0.01M) → 2Al+3(0.01M) + 3Cu(s)
Given: Ecell = 1.98V
The difference between the electrode potentials of two electrodes when no current is drawn through the cell is called ______.
What does the negative sign in the expression `"E"^Θ ("Zn"^(2+))//("Zn")` = − 0.76 V mean?
Assertion: Cu is less reactive than hydrogen.
Reason: `E_((Cu^(2+))/(Cu))^Θ` is negative.
Consider the figure and answer the following question.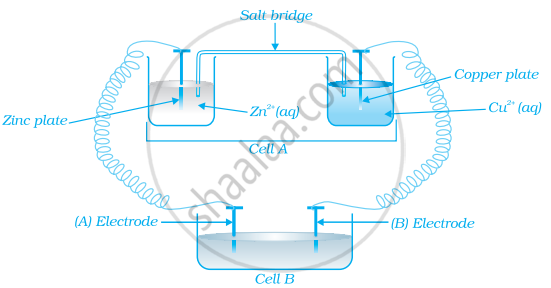
If cell ‘A’ has ECell = 0.5V and cell ‘B’ has ECell = 1.1V then what will be the reactions at anode and cathode?
The standard electrode potential of the two half cells are given below:
\[\ce{Ni^{2+} + 2e^{-} -> Ni, E_0 = - 0.25 Volt}\]
\[\ce{Zn^{2+} + 2e^{-} -> Zn, E_0 = - 0.77 Volt}\]
The voltage of cell formed by combining the two half cells would be?
Which is the correct order of second ionization potential of C, N, O and F in the following?
The potential of a hydrogen electrode at PH = 10 is
A voltaic cell is made by connecting two half cells represented by half equations below:
\[\ce{Sn^{2+}_{ (aq)} + 2e^- -> Sn_{(s)}}\], E0 = − 0.14 V
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\], E0 = + 0.77 V
Which statement is correct about this voltaic cell?
