Advertisements
Advertisements
Question
In the plot of molar conductivity (∧m) vs square root of concentration (c1/2), following curves are obtained for two electrolytes A and B:
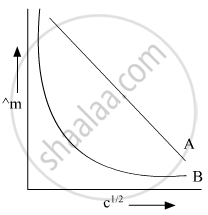
Answer the following:
(i) Predict the nature of electrolytes A and B.
(ii) What happens on extrapolation of ∧m to concentration approaching zero for electrolytes A and B?
Solution
(i)
A = Strong electrolyte
B = Weak electrolyte
(ii) On extrapolation of these graphs they cut the Y-axis it means concentration is approaching zero and at that point, we will say infinite dilution of their respective A and B.
When A and B cut on Y-axis then we will say molar conductivity of A and B at infinite dilution.
APPEARS IN
RELATED QUESTIONS
State Kohlrausch Law
Define limiting molar conductivity.
The molar conductivity of 0.025 mol L−1 methanoic acid is 46.1 S cm2 mol−1. Calculate its degree of dissociation and dissociation constant. Given \[\ce{λ^0_{(H^+)}}\] = 349.6 S cm2 mol−1 and \[\ce{λ^0_{(HCOO^-)}}\] = 54.6 S cm2 mol−1.
Write mathematical expression of molar conductivity of the given solution at infinite dilution.
Calculate the degree of dissociation (α) of acetic acid if its molar conductivity (Λm) is 39.05 S cm2 mol−1.
Given λ°(H+) = 349.6 S cm2 mol−1 and λ°(CH3COO−) = 40.9 S cm2 mol−1
Solutions of two electrolytes ‘A’ and ‘B’ are diluted. The Λm of ‘B’ increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer. Graphically show the behavior of ‘A’ and ‘B’.
Which of the following halogen acids is the strongest reducing agent?
The molar conductance of \[\ce{NaCl, HCl}\] and \[\ce{CH3COONa}\] at infinite dilution are 126.45, 426.16 and 91.0 S cm2 mol−1 respectively. The molar conductance of \[\ce{CH3COOH}\] at infinite dilution is. Choose the right option for your answer.
The unit of molar conductivity is ______.
Discuss the variation of conductivity and molar conductivity with concentration.
