Advertisements
Advertisements
Question
We have 0.5 g of hydrogen gas in a cubic chamber of size 3 cm kept at NTP. The gas in the chamber is compressed keeping the temperature constant till a final pressure of 100 atm. Is one justified in assuming the ideal gas law, in the final state?
(Hydrogen molecules can be consider as spheres of radius 1 Å).
Solution
Assuming hydrogen molecules as spheres of radius 1 Å.
So, r = 1 Å = radius
The volume of hydrogen molecules = `4/3 pir^3`
= `4/3 (3.14)(10^-10)^3`
= `4 xx 10^-30 m^3`
Number of moles of H2 = `"Mass"/"Molecular mass"`
= `0.5/2`
= 0.25
Molecules of H2 present = Number of moles of H2 present × 6.023 × 1023
= 0.25 × 6.023 × 1023
∴ Volume of molecules present = Molecules number × Volume of each molecule
= 0.25 × 6.023 × 1023 × 4 × 10–30
= 6.023 × 1023 × 10–30
= 6 × 10–7 m3 ......(i)
Now, if the ideal gas law is considered to be constant,
`p_iV_i = p_fV_f`
`V_f = (p_i/p_f)`
`V_i = (1/100)(3 xx 10^-2)^3`
= `(27 xx 10^-6)/10^2`
= 2.7 × 10–7 m3 ......(ii)
Hence, on compression, the volume of the gas is of the order of the molecular volume [form equation (i) and equation (ii)]. The intermolecular forces will play a role and the gas will deviate from ideal gas behaviour.
APPEARS IN
RELATED QUESTIONS
The energy of a given sample of an ideal gas depends only on its
Calculate the volume of 1 mole of an ideal gas at STP.
Let Q and W denote the amount of heat given to an ideal gas and the work done by it in an isothermal process.
An ideal gas is taken through a process in which the pressure and the volume are changed according to the equation p = kV. Show that the molar heat capacity of the gas for the process is given by `"C" ="C"_"v" +"R"/2.`
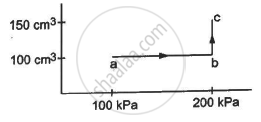
An ideal gas (γ = 1.67) is taken through the process abc shown in the figure. The temperature at point a is 300 K. Calculate (a) the temperatures at b and c (b) the work done in the process (c) the amount of heat supplied in the path ab and in the path bcand (d) the change in the internal energy of the gas in the process.
The volume of an ideal gas (γ = 1.5) is changed adiabatically from 4.00 litres to 3.00 litres. Find the ratio of (a) the final pressure to the initial pressure and (b) the final temperature to the initial temperature.
Consider a given sample of an ideal gas (Cp/Cv = γ) having initial pressure p0 and volume V0. (a) The gas is isothermally taken to a pressure p0/2 and from there, adiabatically to a pressure p0/4. Find the final volume. (b) The gas is brought back to its initial state. It is adiabatically taken to a pressure p0/2 and from there, isothermally to a pressure p0/4. Find the final volume.
Two samples A and B, of the same gas have equal volumes and pressures. The gas in sample A is expanded isothermally to double its volume and the gas in B is expanded adiabatically to double its volume. If the work done by the gas is the same for the two cases, show that γ satisfies the equation 1 − 21−γ = (γ − 1) ln2.
Two vessels A and B of equal volume V0 are connected by a narrow tube that can be closed by a valve. The vessels are fitted with pistons that can be moved to change the volumes. Initially, the valve is open and the vessels contain an ideal gas (Cp/Cv = γ) at atmospheric pressure p0 and atmospheric temperature T0. The walls of vessel A are diathermic and those of B are adiabatic. The valve is now closed and the pistons are slowly pulled out to increase the volumes of the vessels to double the original value. (a) Find the temperatures and pressures in the two vessels. (b) The valve is now opened for sufficient time so that the gases acquire a common temperature and pressure. Find the new values of the temperature and pressure.
The figure shows an adiabatic cylindrical tube of volume V0 divided in two parts by a frictionless adiabatic separator. Initially, the separator is kept in the middle, an ideal gas at pressure p1 and temperature T1 is injected into the left part and another ideal gas at pressure p2 and temperature T2 is injected into the right part. Cp/Cv = γ is the same for both the gases. The separator is slid slowly and is released at a position where it can stay in equilibrium. Find (a) the volumes of the two parts (b) the heat given to the gas in the left part and (c) the final common pressure of the gases.

