Advertisements
Advertisements
Question
What happens when D-glucose is treated with the following reagent?
HI
Solution
When D-glucose is heated with HI for a long time, n-hexane is formed.
\[\begin{array}{cc}
\ce{CHO}\phantom{...............................................}\\
|\phantom{..................................................}\\
\ce{(CHOH)4 ->[HI][\Delta] \underset{{n}-hexane}{CH3 - CH2 - CH2 - CH2 - CH2 - CH3}}\\
|\phantom{..................................................}\\
\ce{\underset{{D-glucose}}{CH2OH}}\phantom{..........................................}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the reaction that indicates the presence of -CHO group in glucose
What happens when glucose is treated with hydroxylamine?
Maltose is a
(a) Polysaccharide
(b) Disaccharide
(c) Trisaccharide
(d) Monosaccharide
What happens when glucose is treated with hydrogen cyanide?
Write the product when D-glucose reacts with conc. HNO3.
The following compound can be called as:
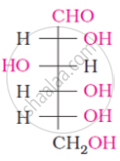
The number of asymmetric carbon atom(s) below the figure is/are

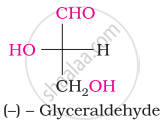
The symbols D and L represents ____________.
Which is the least stable form of glucose?
A solution of D-glucose in water rotates the plane polarised light ____________.
The number of chiral carbon atoms present in cyclic structure α-D(+) glucose:
The α-D glucose and β-D glucose differ from each other due to difference in carbon atom with respect to its ____________.
The two forms of D-glucopyranose obtained from the solution of D-glucose are called ____________.
Choose the correct relationship for glucose and fructose:
Which of the following pairs represents anomers?
Account for the following:
What happens when D – glucose is treated with the following reagents
Bromine water
Consider the following reactions:
(i) \[\ce{Glucose + R-OH ->[Conc. HNO3] [A] ->[X eq of][(CH3CO)2O] Acetyl derivative}\]
(ii) \[\ce{Glucose ->[Ni/H2] [A] ->[Y eq of][(CH3CO)2O] Acetyl derivative}\]
(iii) \[\ce{Glucose ->[Z eq of][(CH3CO)2O] Acetyl derivative}\]
'X, 'Y' and 'Z' in these reactions are respectively:
Give the reaction of glucose with hydrogen cyanide. Presence of which group is confirmed by this reaction?
