Advertisements
Advertisements
Questions
What happens when
H3PO3 is heated ?
Write the reactions involved.
What happens when H3PO3 is heated? Write the equations.
Solution 1
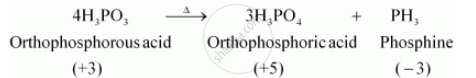
On heating, H3PO3 decomposes into phosphoric acid and phosphine.

Solution 2
H3PO3, on heating, undergoes disproportionation reaction to form PH3 and H3PO4. The oxidation numbers of P in H3PO3, PH3, and H3PO4 are +3, −3, and +5 respectively. As the oxidation number of the same element is decreasing and increasing during a particular reaction, the reaction is a disproportionation reaction.
APPEARS IN
RELATED QUESTIONS
which poisonous gas is evolved when white phosphorus is heated with conc. NaOH solution? Write the chemical equation involved.
How is phosphine prepared using the following reagent ?
HCl
How is phosphine prepared using the following reagent?
H2SO4
How is phosphine prepared using the following reagent?
Caustic soda
Complete the following chemical equations :
Ca3P2 + H2O →
