Advertisements
Advertisements
Question
Why does presence of a nitro group make the benzene ring less reactive in comparison to the unsubstituted benzene ring. Explain.
Solution
Nitro group is an electron-withdrawing group (-R and -I effects). It deactivates the ring by decreasing nucleophilicity for further substitution.
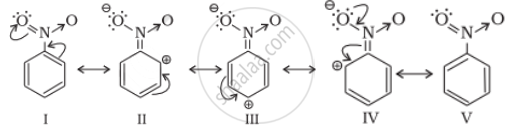
APPEARS IN
RELATED QUESTIONS
Arrange the set of compound in order of their decreasing relative reactivity with an electrophile, E+ Chlorobenzene, 2,4-dinitrochlorobenzene, p-nitrochlorobenzene.
Arrange the set of compound in order of their decreasing relative reactivity with an electrophile, E+ Toluene, p-H3C–C6H4–NO2, p-O2N–C6H4–NO2.
Out of benzene, m–dinitrobenzene and toluene which will undergo nitration most easily and why?
Suggest the name of a Lewis acid other than anhydrous aluminium chloride which can be used during ethylation of benzene.
For an electrophilic substitution reaction, the presence of a halogen atom in the benzene ring:
(i) deactivates the ring by inductive effect
(ii) deactivates the ring by resonance
(iii) increases the charge density at ortho and para position relative to meta position by resonance
(iv) directs the incoming electrophile to meta position by increasing the charge density relative to ortho and para position.
In an electrophilic substitution reaction of nitrobenzene, the presence of nitro group:
(i) Deactivates the ring by inductive effect.
(ii) Activates the ring by inductive effect.
(iii) Decreases the charge density at ortho and para position of the ring relative to meta position by resonance.
(iv) Increases the charge density at meta position relative to the ortho and para positions of the ring by resonance.
Assertion (A): Nitration of benzene with nitric acid requires the use of concentrated sulphuric acid.
Reason (R): The mixture of concentrated sulphuric acid and concentrated nitric acid produces the electrophile, \[\ce{NO^{+}2}\].
In Friedel-Crafts alkylation of aniline, one gets ______.
Excess of isobutane on reactions with Br2 in presence of light at 125°C gives which one of the following, as the major product?
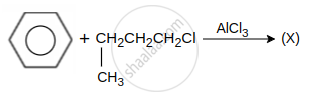
hydrocarbon (X) major product X is:
