Advertisements
Advertisements
Question
Why is Sc3+ colourless while Ti3+ coloured? (Atomic number Sc = 21, Ti =22)
Solution
Ti3+ is a 3d1 system. The colour is due to d–d transition. There is one d electron present in the 3d subshell. When light falls on the Ti3+ complex, the t2g electron is excited to the eg level. This excitation takes place in the greenish yellow region (≈5000 A°) and the rest is transmitted. The complementary colour is transmitted which is violet.
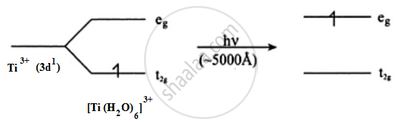
Sc3+ is a 3d0 system. There are no d electrons; hence, d–d transition is not possible. Hence, Sc3+ is colourless.
APPEARS IN
RELATED QUESTIONS
What are interstitial compounds?
Explain why is Fe3+ more stable than Fe2+?
Account for the following:
Mn shows the highest oxidation state of +7 with oxygen but with fluorine, it shows oxidation state of +4.
Why do the transition elements have higher enthalpies of atomisation?
How would you account for the following: Transition metals form complex compounds.
How would you account for the following : Transition metals form coloured compounds
Comment on the statement that elements of the first transition series possess many properties different from those of heavier transition elements.
An antifriction alloy made up of antimony with tin and copper, which is extensively used in machine bearings is called _______.
(A) Duralumin
(B) Babbitt metal
(C) Spiegeleisen
(D) Amalgam
How would you account for the following?
Transition metals and their compounds act as catalysts.
Give reasons Iron has the higher enthalpy of atomization than that of copper.
Account for the following :
Ti4+ is colourless whereas V4+ is coloured in an aqueous solutions.
Why do transition metals exhibit higher enthalpy of atomization?
Explain why transition elements form alloys.
Transition elements show magnetic moment due to spin and orbital motion of electrons. Which of the following metallic ions have almost same spin only magnetic moment?
(i) \[\ce{Co^{2+}}\]
(ii) \[\ce{Cr^{2+}}\]
(iii) \[\ce{Mn^{2+}}\]
(iv) \[\ce{Cr^{3+}}\]
Why EΘ values for Mn, Ni and Zn are more negative than expected?
Ionisation enthalpies of Ce, Pr and Nd are higher than Th, Pa and U. Why?
The halides of transition elements become more covalent with increasing oxidation state of the metal. Why?
Match the properties given in Column I with the metals given in Column II.
| Column I (Property) | Column II (Metal) | |
| (i) | Element with highest second ionisation enthalpy |
(a) \[\ce{Co}\] |
| (ii) | Element with highest third ionisation enthalpy |
(b) \[\ce{Cr}\] |
| (iii) | \[\ce{M}\] in \[\ce{M(CO)6}\] is | (c) \[\ce{Cu}\] |
| (iv) | Element with highest heat of atomisation |
(d) \[\ce{Zn}\] |
| (e) \[\ce{Ni}\] |
Transition metals can act as catalysts because these can change their oxidation state. How does \[\ce{Fe(III)}\] catalyse the reaction between iodide and persulphate ions?
Catalytic hydrogenation of benzene gives
Which does not belong to first transition series?
Photographic film and plates have - au essential ingredient of
The basic character of transition metals monoxide follow the order.
Why Zn, Cd and Hg are not called transition metals?
Which of the following ions acts as a typical transition metal ion?
Assertion (A): Transition metals have high enthalpy of atomisation.
Reason (R): Greater number of unpaired electrons in transition metals results in weak metallic bonding.
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Atomic sizes
