Advertisements
Advertisements
प्रश्न
Why is Sc3+ colourless while Ti3+ coloured? (Atomic number Sc = 21, Ti =22)
उत्तर
Ti3+ is a 3d1 system. The colour is due to d–d transition. There is one d electron present in the 3d subshell. When light falls on the Ti3+ complex, the t2g electron is excited to the eg level. This excitation takes place in the greenish yellow region (≈5000 A°) and the rest is transmitted. The complementary colour is transmitted which is violet.
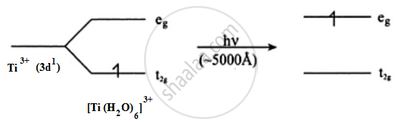
Sc3+ is a 3d0 system. There are no d electrons; hence, d–d transition is not possible. Hence, Sc3+ is colourless.
APPEARS IN
संबंधित प्रश्न
What are interstitial compounds?
Why do interstitial compounds have higher melting points than corresponding pure metals?
Complete the following chemical equations:
`(i) Cr_2O_7^(2-)+6Fe^(2+)+14H^+ ->`
`(ii) 2CrO_4^(2-)+2H^+ ->`
`(iii) 2MnO_4^-+5C_2O_4^(2-)+16H^+ ->`
Why +2 oxidation state of manganese is more stable?
The elements of 3d transition series are given as: Sc Ti V Cr Mn Fe Co
Answer the following: Write the element which shows maximum number of oxidation states. Give reason.
The elements of 3d transition series are given as: Sc Ti V Cr Mn Fe Co
Answer the following: Which element has the highest m.p?
What are the transition elements? Write two characteristics of the transition elements.
How would you account for the irregular variation of ionization enthalpies (first and second) in the first series of the transition elements?
Why are Mn2+ compounds more stable than Fe2+ towards oxidation to their +3 state?
Which of the d-block elements may not be regarded as the transition elements?
What are alloys?
NF3 is possible, but NF5 is not. Why?
Why do transition metal ions possess a great tendency to form complexes?
The paramagnetic character in-3d-transition series elements increases-: up to Mn and then decreases.
Give reasons for the following:
The transition metals generally form coloured compounds.
In lake test for Al3+ ions, there is the formation of coloured ‘floating lake’. It is due to ______.
Read the passage given below and answer the following question:
The transition metals when exposed to oxygen at low and intermediate temperatures form thin, protective oxide films of up to some thousands of Angstroms in thickness. Transition metal oxides lie between the extremes of ionic and covalent binary compounds formed by elements from the left or right side of the periodic table. They range from metallic to semiconducting and deviate by both large and small degrees from stoichiometry. Since electron bonding levels are involved, the cations exist in various valence states and hence give rise to a large number of oxides. The crystal structures are often classified by considering a cubic or hexagonal close-packed lattice of one set of ions with the other set of ions filling the octahedral or tetrahedral interstices. The actual oxide structures, however, generally show departures from such regular arrays due in part to distortions caused by packing of ions of different size and to ligand field effects. These distortions depend not only on the number of d-electrons but also on the valence and the position of the transition metal in a period or group.
In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
Assertion: Cations of transition elements occur in various valence states.
Reason: Large number of oxides of transition elements are possible.
Although Zirconium belongs to 4d transition series and Hafnium to 5d transition series even then they show similar physical and chemical properties because ______.
Why does copper not replace hydrogen from acids?
Out of \[\ce{Cu2Cl2}\] and \[\ce{CuCl2}\], which is more stable and why?
Identify the metal and justify your answer.
\[\ce{MO3F}\]
Mention any three processes where transition metals act as catalysts.
A violet compound of manganese (A) decomposes on heating to liberate oxygen and compounds (B) and (C) of manganese are formed. Compound (C) reacts with KOH in the presence of potassium nitrate to give compound (B). On heating compound (C) with conc. \[\ce{H2SO4}\] and \[\ce{NaCl}\], chlorine gas is liberated and a compound (D) of manganese along with other products is formed. Identify compounds A to D and also explain the reactions involved.
Give reasons for the following statement:
\[\ce{Zn}\], \[\ce{Cd}\] and \[\ce{Hg}\] are soft metals.
Consider the following standard electrode potential values:
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\], E0 = +0.77 V
\[\ce{MnO^{-4}_{ (aq)} + 8H^+ + 5e^- -> Mn^{2+}_{ (aq)} + 4H2O_{(l)}}\], E0 = +1.51 V
What is the cell potential for the redox reaction?
What is the oxidation state of chromium in chromate ion and dichromate ion?
Explain the magnetic properties of d-block (or transition) elements.
Describe the oxidising action of potassium dichromate and write the ionic equation for its reaction with iron (II) solution.
Decide which of the following atomic numbers are the atomic numbers of the inner transition elements:
29, 59, 74, 95, 102, 104
