Advertisements
Advertisements
प्रश्न
Why is Sc3+ colourless while Ti3+ coloured? (Atomic number Sc = 21, Ti =22)
उत्तर
Ti3+ is a 3d1 system. The colour is due to d–d transition. There is one d electron present in the 3d subshell. When light falls on the Ti3+ complex, the t2g electron is excited to the eg level. This excitation takes place in the greenish yellow region (≈5000 A°) and the rest is transmitted. The complementary colour is transmitted which is violet.
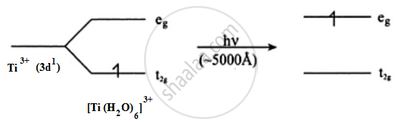
Sc3+ is a 3d0 system. There are no d electrons; hence, d–d transition is not possible. Hence, Sc3+ is colourless.
APPEARS IN
संबंधित प्रश्न
How would you account for the following : Transition metals form coloured compounds
Which is a stronger reducing agent, Cr2+ or Fe2+ and why?
What may be the stable oxidation state of the transition element with the following d electron configurations in the ground state of their atoms?
3d3, 3d5, 3d8 and 3d4
Compare the stability of +2 oxidation state for the elements of the first transition series.
How would you account for the following:
The d1 configuration is very unstable in ions.
Calculate the number of unpaired electrons in the following gaseous ions:
Mn3+, Cr3+, V3+ and Ti3+. Which one of these is the most stable in an aqueous solution?
Use Hund’s rule to derive the electronic configuration of Ce3+ ion and calculate its magnetic moment on the basis of ‘spin-only’ formula.
Write down the number of 3d electrons in the following ion:
Cu2+
Indicate how would you expect the five 3d orbitals to be occupied for this hydrated ions (octahedral).
Following are the transition metal ions of 3d series:
Ti4+, V2+, Mn3+, Cr3+
(Atomic numbers: Ti = 22, V = 23, Mn = 25, Cr = 24)
Answer the following:
1) Which ion is most stable in an aqueous solution and why?
2) Which ion is a strong oxidising agent and why?
3) Which ion is colourless and why?
How would you account for the following?
Zr (Z = 40) and Hf (Z = 72) have almost identical radii.
Give reasons: Sc3+ is colourless in aqueous solution whereas Ti3+ is coloured.
Why does the density of transition elements increase from Titanium to Copper? (at. no. Ti = 22,
Cu = 29)
Why do transition metal ions possess a great tendency to form complexes?
When \[\ce{KMnO4}\] solution is added to oxalic acid solution, the decolourisation is slow in the beginning but becomes instantaneous after some time because ______.
Match the catalysts given in Column I with the processes given in Column II.
| Column I (Catalyst) | Column II (Process) |
| (i) \[\ce{Ni}\] in the presence of hydrogen | (a) Zieglar Natta catalyst |
| (ii) \[\ce{Cu2C12}\] | (b) Contact process |
| (iii) \[\ce{V2O5}\] | (c) Vegetable oil to ghee |
| (iv) Finely divided iron | (d) Sandmeyer reaction |
| (v) \[\ce{TiCl4 + Al (CH3)3}\] | (e) Haber's Process |
| (f) Decomposition of KCIO3 |
Match the properties given in Column I with the metals given in Column II.
| Column I (Property) | Column II (Metal) | |
| (i) | An element which can show +8 oxidation state | (a) \[\ce{Mn}\] |
| (ii) | 3d block element that can show | (b) \[\ce{Cr}\] |
| upto +7 oxidation state | (c) \[\ce{Os}\] | |
| (iii) | 3d block element with highest melting point | (d) \[\ce{Fe}\] |
Assertion: Separation of \[\ce{Zr}\] and \[\ce{Hf}\] is difficult.
Reason: Because \[\ce{Zr}\] and \[\ce{Hf}\] lie in the same group of the periodic table.
The element with atomic number 53 belongs to
On strong heating AgNO3, the gases evolved are:-
Which of the following species has maximum magnetic momentum?
Which of the following transition metal is not coloured?
Give reason for the following statement:
Physical and chemical properties of the 4d and 5d series of the transition elements are quite similar to expected.
The number of terminal oxygen atoms present in the product B obtained from the following reactions is:
\[\ce{FeCr2O4 + Na2CO3 + O2 -> A + Fe2O3 + CO2}\]
\[\ce{A + H^+ -> B + H2O + Na^+}\]
The disproportionation of \[\ce{MnO^{2-}_4}\] in acidic medium resulted in the formation of two manganese compounds A and B. If the oxidation state of Mn in B is smaller than that of A, then the spin-only magnetic moment (µ) value of B in BM is ______. (Nearest integer)
Consider the following standard electrode potential values:
\[\ce{Sn^{2+}_{ (aq)} + 2e^- -> Sn_{(s)}}\]; E0 = −0.14 V
\[\ce{Fe^{3+}_{ (aq)} + e^- -> Fe^{2+}_{ (aq)}}\]; E0 = +0.77 V
What is the cell reaction and potential for the spontaneous reaction that occurs?
Write the number of unpaired electrons in Cr3+.
(Atomic number of Cr = 24)
Give a reason for the following:
Transition metals possess a great tendency to form complex compounds.
Account for the following:
Zirconium (Zr) and Hafnium (Hf) are difficult to separate.
Describe the oxidising action of potassium dichromate and write the ionic equation for its reaction with iron (II) solution.
