Advertisements
Advertisements
Question
Write a short note on diazotisation.
Solution 1
Benzenediazonium chloride is prepared by the reaction of aniline with nitrous acid at 273-278K. Nitrous acid is produced in the reaction mixture by the reaction of sodium nitrite with hydrochloric acid. The conversion of primary aromatic amines into diazonium salts is known as diazotisation. Due to its instability, the diazonium salt is not generally stored and is used immediately after its preparation.
\[\ce{C6H5NH2 + NaNO2 + 2HCl ->[273 - 278 K] C6H5\overset{+}{N2}\overset{-}{C}l + NaCl + 2H2O}\]
Solution 2
Aniline reacts with nitrous acid at low temperature (273 – 278 K) to give benzene diazonium chloride which is stable for a short time and slowly decomposes even at low temperatures. This reaction is known as diazotization.

RELATED QUESTIONS
The conversion of primary aromatic amines into diazonium salts is known as ___________
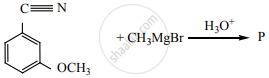
Product ‘P’ in the above reaction is:
Ammonium salt of benzoic acid is heated strongly with P2O5 and the product so formed is reduced and then treated with NaNO2/HCl at low temperature. The final compound formed is ____________.
Identify X in the sequence given below
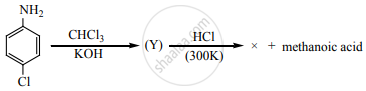
The major product of the following reaction:
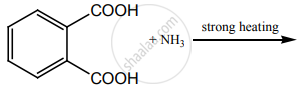
Write a short note on the following.
Coupling reaction
Write a short note on the following.
Gomberg reaction
Account for the following.
Diazonium salts of aromatic amines are more stable than those of aliphatic amines.
Account for the following.
Amines are more basic than amides.
Identify A, B, C and D.
\[\ce{aniline + benzaldehyde -> A ->[Conc. HNO3][B] C + D}\]
Why is \[\ce{NH2}\] group of aniline acetylated before carrying out nitration?
What would be the major product of the following reaction?
\[\ce{C6H5 - CH2 - OC6H5 + HBr -> A + B}\]
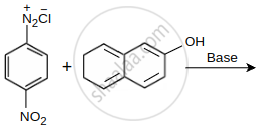
Coupling of benzene diazonium chloride with 1-naphthol in alkaline medium will give:
The major product of the following reaction is: