Advertisements
Advertisements
Question
(a) Write the basic nuclear process involved in the emission of β+ in a symbolic form, by a radioactive nucleus.
(b) In the reactions given below:
(i)`""_16^11C->_y^zB+x+v`
(ii)`""_6^12C+_6^12C->_a^20 Ne + _b^c He`
Find the values of x, y, and z and a, b and c.
Solution
(a)
The basic nuclear process involved in the emission of β+ in a symbolic form, by a radioactive nucleus
p →n + β++ v
For a beta-plus decay, a proton transforms into a neutron within the nucleus, according to the above reaction.
(b)
(i)
`""_6^11C->_y^zB+x+v`
`=>""_6^11C->_5^11B+_1^0beta+100`
The corresponding y and z are 5 and 11, respectively. The x is the positron
(ii)
`""_6^12C+_6^12C->_a^20 Ne +_b^c He`
⇒
`""_6^12C+_6^12C->_10^20Ne+_2^4He`
The corresponding values of a, b and c are 10, 2 and 4, respectively.
APPEARS IN
RELATED QUESTIONS
Derive the mathematical expression for law of radioactive decay for a sample of a radioactive nucleus
A radioactive isotope has a half-life of T years. How long will it take the activity to reduce to a) 3.125%, b) 1% of its original value?
Obtain the amount of `""_27^60"Co"` necessary to provide a radioactive source of 8.0 mCi strength. The half-life of `""_27^60"Co"` is 5.3 years.
Represent Radioactive Decay curve using relation `N = N_o e^(-lambdat)` graphically
A radioactive nucleus 'A' undergoes a series of decays as given below:
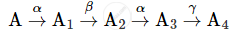
The mass number and atomic number of A2 are 176 and 71 respectively. Determine the mass and atomic numbers of A4 and A.
(a) Derive the relation between the decay constant and half life of a radioactive substance.
(b) A radioactive element reduces to 25% of its initial mass in 1000 years. Find its half life.
A radioactive nucleus ‘A’ undergoes a series of decays according to the following scheme:
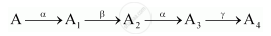
The mass number and atomic number of A are 180 and 72 respectively. What are these numbers for A4?
What is the amount of \[\ce{_27^60Co}\] necessary to provide a radioactive source of strength 10.0 mCi, its half-life being 5.3 years?
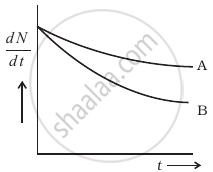
Which sample, A or B shown in figure has shorter mean-life?
A piece of wood from the ruins of an ancient building was found to have a 14C activity of 12 disintegrations per minute per gram of its carbon content. The 14C activity of the living wood is 16 disintegrations per minute per gram. How long ago did the tree, from which the wooden sample came, die? Given half-life of 14C is 5760 years.
