Advertisements
Advertisements
Question
Write the structures of the main products of the following reactions:

Solution
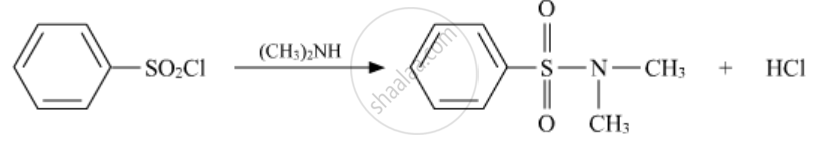
APPEARS IN
RELATED QUESTIONS
Arrange the following in increasing order of their basic strength in aqueous solution:
\[\ce{CH3NH2, (CH3)3N, (CH3)2NH}\]
Why does NH3 act as a Lewis base?
Arrange the following:
In increasing order of basic strength:
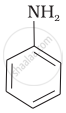
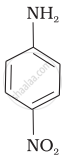
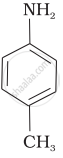
Aniline, p-nitroaniline and p-toluidine
The correct increasing order of basic strength for the following compounds is ______.
(I)
(II)
(III)
The correct decreasing order of basic strength of the following species is ______.
\[\ce{H2O, NH3, OH-, NH^{-}2}\]
By the presence of a halogen atom in the ring, what is the effect of this on basic property of aniline?
What is the correct decreasing order of the basic character of the three amines and ammonia?
Give reasons for the following observation:
pKb of aniline is lower than the m-nitroaniline.
Which of the following compound cannot be produced if 1-propane amine is treated with NaNO2 and HCl?
State one reason for the following:
Alkylamine is soluble in water, whereas arylamine is insoluble in water.
