Advertisements
Advertisements
Question
State one reason for the following:
Alkylamine is soluble in water, whereas arylamine is insoluble in water.
Solution
Alkylamine is soluble in water because it has the potential to form hydrogen bonds with water. On the other hand, arylamines contain benzene, which is very hydrophobic. As a result, arylamines cannot be dissolved in water.
APPEARS IN
RELATED QUESTIONS
Write the chemical equations involved when aniline is treated with the following reagents: HCI
Arrange the following:
In decreasing order of basic strength in the gas phase:
C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3
The correct increasing order of basic strength for the following compounds is ______.
(I)
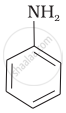
(II)
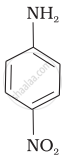
(III)
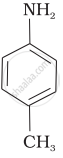
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
Account for the following:
Acylation of aniline is carried out in the presence of pyridine.
When methyl iodide is heated with ammonia, what is the product obtained?
By the presence of a halogen atom in the ring, what is the effect of this on basic property of aniline?
What is the characteristic smell of liquid amines?
Give reasons for the following observation:
pKb of aniline is lower than the m-nitroaniline.
Account for the following:
Arrange the following compounds in the increasing order of their basic strength in aqueous solution: CH3NH2,(CH3)3N,(CH3)2NH
