SSC (English Medium)
SSC (Marathi Semi-English)
Academic Year: 2024-2025
Date: March 2025
Advertisements
General Instructions :
- All questions are compulsory.
- Use of a calculator is not allowed.
- The numbers to the right of the questions indicate fu ll marks.
- In case of MCQs (Q. No. 1(A)) only the first attempt will be evaluated and will be given credit.
- For each MCQ, the correct alternative (A), (B), (C), or (D) with a subquestion number is to be written as an answer.
For Eg. : (i) (A), (ii) (B), (iii) (C) - Scientifically correct, labelled diagrams should be drawn wherever necessary.
A Rainbow is a beautiful natural phenomenon formed because of the following:
Dispersion of light
Refraction of light
Total internal reflection of light
All of these
Chapter:
Which of the following statement is incorrect with reference to the reaction of silver nitrate with sodium chloride?
Chemical formula for silver nitrate is AgNO3.
Silver nitrate on reaction with sodium chloride gives white precipitate.
Precipitate formed is soluble in nature.
When aqueous solutions of the two are mixed a double replacement reaction takes place.
Chapter: [0.03] Chemical Reactions and Equations
The moon attracts Earth with a force of 1020 N, and then the force with which the Earth attracts the moon is ______.
Less than 1020 N.
1020 N.
10−20 N.
greater than 1020 N.
Chapter: [0.01] Gravitation
The distance between the optical centre and principal focus of a lens is known as ______.
focal length
radius of curvature
optical centre
principle axis
Chapter: [0.07] Lenses
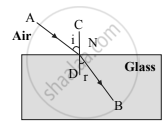
In refraction of light through a glass slab, the directions of the incident ray and the refracted ray are ______.
Perpendicular to each other.
Non-parallel to each other.
Parallel to each other.
Intersecting each other.
Chapter: [0.06] Refraction of Light [0.17] Wonders of Light 2
Find the odd one out:
Lithium (Li)
Sodium (Na)
Nitrogen (N)
Magnesium (Mg)
Chapter: [0.08] Metallurgy
Complete the correlation:
IRNSS : Direction showing : : INSAT : ______ Satellite
Chapter: [0.1] Space Missions
Write true or false:
The anomalous behaviour of water can be studied with Hope's apparatus.
True
False
Chapter:
Match the columns:
| Column ‘A’ | Column ‘B’ |
| Manganese | (a) Metal |
| (b) Non-metal | |
| (c) Transition metal |
Chapter: [0.02] Periodic Classification of Elements
How much is the relative humidity at the dew point?
Chapter: [0.05] Heat
Advertisements
Give scientific reasons.
Silver amalgam is used for filling dental cavities.
Chapter: [0.03] Chemical Reactions and Equations [0.08] Metallurgy
In the electric equipment producing heat like iron, electric heater, boiler, toaster etc., an alloy such as nichrome is used, not pure metals.
Chapter: [0.04] Effects of Electric Current [0.14] The Electric Spark
Give scientific reasons for the following:
Newton's gravitational law is the universal law of gravitation.
Chapter: [0.01] Gravitation
Write the IUPAC name for the following structural formula:
\[\begin{array}{cc}
\ce{CH3-CH-CH3}\\
|\phantom{..}\\
\ce{OH}
\end{array}\]
Chapter:
Observe the following structure and give IUPAC name for the structural formula correctly.
\[\begin{array}{cc}
\ce{O}\phantom{..............}\\
||\phantom{..............}\\
\ce{CH3 - C - CH2 - CH2 - CH3}
\end{array}\]
Chapter:
Write a short note on Presbyopia.
Chapter: [0.07] Lenses
Observe the given figure of a current-carrying solenoid and write the labels of A and B correctly.
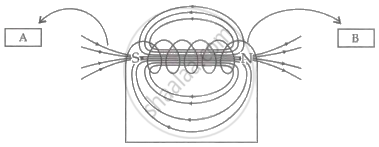
Chapter: [0.04] Effects of Electric Current [0.15] All about Electromagnetism
How much heat energy is necessary to raise the temperature of 5 kg of water from 20°C to 100°C?
Chapter: [0.05] Heat
- Complete the given chemical reaction:
\[\ce{CuSO4_{(aq)} + Fe_{(s)} ->}\] _______ + _______ - Name the type of the reaction.
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
Advertisements
Answer the following questions related to the modern periodic table.
Name the blocks with the groups that they consist of.
Chapter: [0.02] Periodic Classification of Elements
Answer the following questions related to the modern periodic table.
Which block elements are known as transition elements?
Chapter: [0.02] Periodic Classification of Elements
Answer the following questions related to the modern periodic table.
How are elements placed around the zig-zag line in the p-block of the periodic table?
Chapter: [0.02] Periodic Classification of Elements
The figure given below shows the magnetic field around the conductor. Study it and answer the questions:

- What changes are caused by increasing current?
- What do you see when the magnetic needle is kept a little away from the wire?
- In the second part of the figure, the iron filings arrange themselves in a circular manner around the wire. Why does this happen?
Chapter: [0.04] Effects of Electric Current
Observe the given figure and answer the following questions:
- Name the process represented by the figure.
- State the two laws related to the process.
Chapter:
State the importance of Space Missions.
Chapter: [0.1] Space Missions
Observe the given figure showing the orbit of a planet moving around the Sun and write the three laws related to it:

The orbit of a planet moving around the Sun
Chapter: [0.01] Gravitation
Read the text below and answer the questions that follow:
A small amount of hydrochloric acid was taken in a test tube. The test tube was heated. A glass rod was dipped in the ammonia solution and held on the top of the test tube. A white smoke was seen emanating from the tip of the glass rod.
- What must have happened?
- Which colour of gas is formed?
- Write the chemical equation for the reaction.
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
Currently, there are more than 2,500 man-made satellites orbiting the earth. The satellites are launched with a specific objective in mind pertaining to several uses.
Answer the following questions based on the concept of satellites given in the passage:
- Name a natural satellite of the earth.
- What is an artificial satellite? Name the first artificial satellite.
- What is the purpose of sending such satellites?
Chapter: [0.1] Space Missions
Complete the following table for convex lens:
| Sr. No. | Position of the object | Position of the image | Nature of the image |
| 1. | Beyond 2F1 | __________________ | ________________________ |
| 2. | __________________ | At infinity | ________________________ |
| 3. | __________________ | __________________ | Real, inverted and enlarged |
Chapter: [0.16] Wonders of Light 1
Observe the following diagram and answer the questions.

- Construction of which equipment does the above diagram show?
- On which principle does this equipment work?
- According to which law the coil ABCD rotates?
- Write the law in your own words.
- Where is this equipment used?
Chapter: [0.04] Effects of Electric Current [0.15] All about Electromagnetism
Observe the following figure and write the answer of the question.

- Which process is shown in the figure?
- Explain the chemical reaction shown in the figure.
- Write the reactions on anode and cathode.
Chapter: [0.03] Chemical Reactions and Equations [0.08] Metallurgy
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 10th Standard Board Exam Science and Technology 1 with solutions 2024 - 2025
Previous year Question paper for Maharashtra State Board 10th Standard Board Exam -2025 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science and Technology 1, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 10th Standard Board Exam.
How Maharashtra State Board 10th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Science and Technology 1 will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
