Advertisements
Advertisements
प्रश्न
Define the following terms:
Half-life period of reaction (t1/2).
उत्तर
For zero-order reaction,
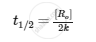
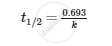
APPEARS IN
संबंधित प्रश्न
In a first order reaction x → y, 40% of the given sample of compound remains unreacted in 45 minutes. Calculate rate constant of the reaction.
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
For a general reaction A → B, plot of concentration of A vs time is given in figure. Answer the following question on the basis of this graph.
(i) What is the order of the reaction?
(ii) What is the slope of the curve?
(iii) What are the units of rate constant?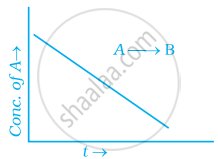
Why is the probability of reaction with molecularity higher than three very rare?
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
A catalyst in a reaction changes which of the following?
The role of a catalyst is to change
In the presence of a catalyst, the heat evolved or absorbed during the reaction.
The number of molecules of the reactants taking part in a single step of the reaction is indicative of ______.
