Advertisements
Advertisements
प्रश्न
Explain the following reaction with their balanced chemical equation.
Ammonia gas reacts with hydrogen chloride.
उत्तर
Ammonia gas and hydrogen chloride gas react to form the salt ammonium chloride. It is a combination reaction.
\[\ce{\underset{\text{Ammonia}}{NH_{3(g)}} + \underset{\text{Hydrogen chloride}}{HCl_{(g)}} -> \underset{\text{Ammonium chloride}}{NH4Cl_{(s)}}}\]
संबंधित प्रश्न
A solution of a substance ‘X’ is used for white washing.
- Name the substance ‘X’ and write its formula.
- Write the reaction of the substance ‘X’ named in (i) above with water.
Why is respiration considered an exothermic reaction? Explain.
When crystals of copper sulphate are strongly heated, the residue obtained is ___________.
(A) red in colour
(B) blue in colour
(C) green in colour
(D) colourless
What happens chemically when quicklime is added to water filled in a bucket?
One of the following is an endothermic reaction. This is:
(a) combination of carbon and oxygen to form carbon monoxide
(b) combination of nitrogen and oxygen to form nitrogen monoxide
(c) combination of glucose and oxygen to form carbon dioxide and water
(d) combination of zinc and hydrochloric acid to form zinc chloride and hydrogen
A silvery-white metal X taken in the form of ribbon, when ignited, burns in air with a dazzling white flame to form a white powder Y. When water is added to powder Y, it dissolves partially to form another substance Z.
(a) What could metal X be?
(b) What is powder Y?
(c) With which substance metal X combines to form powder Y?
(d) What is substance Z? Name one domestic use of substance Z.
(e) Write a balanced chemical equation of the reaction which takes place when metal X burns in air to form powder Y.
What type of chemical reaction take place when ammonia and hydrogen chloride are mixed?
Give one example of a combination reaction which is also exothermic.
Give one example of a combination reaction which is also endothermic.
Consider the following equation of the chemical reaction of a metal M:
4M + 3O2 → 2M2O3
This equation represents:
(a) combination reaction as well as reduction reaction
(b) decomposition reaction as well as oxidation reaction
(c) oxidation reaction as well as displacement reaction
(d) combination reaction as well as oxidation reaction
When hydrogen burns in oxygen, water is formed and when water is electrolysed, then hydrogen and oxygen are produced. What type of reaction takes place:
(a) in the first case?
(b) in the second case
Define a chemical reaction.
What do you understand by a chemical bond ?
Classify the following reaction as combination, decomposition, displacement, precipitation and neutralization. Also balance the equation.
\[\ce{CaCO3_{(s)} ->[heat]CaO_{(s)} + CO2_{(g)}}\]
Explain the following type of chemical reaction, giving two examples for it:
Combination reaction
What are chemical combination or synthesis reactions ? Chemical combination or synthesis:
What effect does an increase in concentration of H+ (aq.) in a solution have on the pH of solution?
Explain the terms with examples.
Combination reaction
Define: Chemical combination reaction
Calcium oxide reacts vigorously with water to produce slaked lime.
\[\ce{CaO{(s)} + H2O(l) -> Ca(OH)2(aq)}\]
This reaction can be classified as:
(A) Combination reaction
(B) Exothermic reaction
(C) Endothermic reaction
(D) Oxidation reaction
Classify the following reaction into different type:
CaO(s) + H2O(l) → Ca(OH)2(aq)
State why a direct combination reaction is called a – ‘synthesis reaction’.
Give a balanced equation for –
A direct combination reaction involving two elements, one of which is a non-metal
Select the correct answer for the statement given below:
The product formed during direct combination reaction of carbon dioxide and water.
Complete the statement by filling in the blank with the correct word:
Direct combination reaction of phosphorus pentoxide with water gives _______.
Which changes occur during chemical changes?
H2(g) + Cl29(g) → 2HCl(g) is a ______
Which of the following is not a physical change?
The respiration process during which glucose undergoes slow combustion by combining with oxygen in the cells of our body to produce energy is a kind of:
The Salt Story
From: The New Indian Express 9 March 2021
The salt pans in Marakkanam, a port town about 120 km from Chennai are the third largest producer of salt in Tamil Nadu. Separation of salt from water is a laborious process and the salt obtained is used as raw materials for manufacture of various sodium compounds.
One such compound is Sodium hydrogen carbonate, used in baking, as an antacid and in soda acid fire extinguishers.
The table shows the mass of various compounds obtained when 1litre of sea water is evaporated.
| COMPOUND | FORMULA | MASS OF SOLID PRESENT /g |
| Sodium Chloride | NaCl | 28.0 |
| Magnesium Chloride | MgCl2 | 8.0 |
| Magnesium Sulphate | MgSO4 | 6.0 |
| Calcium Sulphate | CaSO4 | 2.0 |
| Calcium Carbonate | CaCO3 | 1.0 |
| TOTAL AMOUNT OF SALT OBTAINED | 45.0 | |
Which compound in the table reacts with acids to release carbon dioxide?
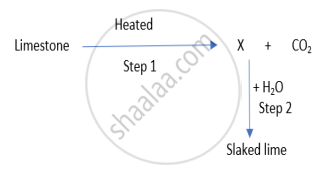
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Identify the types of chemical reactions occurring during the combustion of fuel:
Balance the following chemical equation and identify the type of chemical reaction.
`"Mg"("s") + "Cl"_2("g") -> "MgCl"_2("s")`
A clear solution of slaked lime is made by dissolving Ca(OH)2 in an excess of water. This solution is left exposed to air. The solution slowly goes milky as a faint white precipitate forms. Explain why a faint white precipitate forms, support your response with the help of a chemical equation.
