Advertisements
Advertisements
प्रश्न
When crystals of copper sulphate are strongly heated, the residue obtained is ___________.
(A) red in colour
(B) blue in colour
(C) green in colour
(D) colourless
उत्तर
colourless
APPEARS IN
संबंधित प्रश्न
Find the odd man out:
Camphor, Ammonium Chloride, Naphthalene balls, Sugar
A silvery-white metal X taken in the form of ribbon, when ignited, burns in air with a dazzling white flame to form a white powder Y. When water is added to powder Y, it dissolves partially to form another substance Z.
(a) What could metal X be?
(b) What is powder Y?
(c) With which substance metal X combines to form powder Y?
(d) What is substance Z? Name one domestic use of substance Z.
(e) Write a balanced chemical equation of the reaction which takes place when metal X burns in air to form powder Y.
Gas A, which is the major cause of global warming, combines with hydrogen oxide B in nature in the presence of an environmental factor C and a green material D to form a six carbon organic compounds E and a gas F. The gas F is necessary for breathing.
(a) What is gas A?
(b) What is the common name of B?
(c) What do you think could be C?
(d) What is material D? Where is it found?
(e) Name the organic compound E.
(f) What is gas F? Name the natural process during which it is released.
What type of reaction is represented by the following equation?
2 Ca + O2 → 2CaO
What type of chemical reaction is represented by the following equation?
A + B → C
What type of reaction is represented by the following equation?
CaO + CO2 → CaCO3
Give one example of a combination reaction which is also endothermic.
When hydrogen burns in oxygen, water is formed and when water is electrolysed, then hydrogen and oxygen are produced. What type of reaction takes place:
(a) in the first case?
(b) in the second case
What happens during a chemical reaction ?
What do you observe when solid lead nitrate is heated?
What do you observe when when dilute sulphuric acid is added to barium chloride solution ?
Explain the following type of chemical reaction, giving two examples for it:
Combination reaction
What are chemical combination or synthesis reactions ? Chemical combination or synthesis:
What effect does an increase in concentration of H+ (aq.) in a solution have on the pH of solution?
Calcium oxide reacts vigorously with water to produce slaked lime.
\[\ce{CaO{(s)} + H2O(l) -> Ca(OH)2(aq)}\]
This reaction can be classified as:
(A) Combination reaction
(B) Exothermic reaction
(C) Endothermic reaction
(D) Oxidation reaction
Classify the following reaction into different type:
CaO(s) + H2O(l) → Ca(OH)2(aq)
State why a direct combination reaction is called a – ‘synthesis reaction’.
Give a balanced equation for –
A direct combination reaction involving two elements, one of which is a non-metal
Complete the statement by filling in the blank with the correct word:
Direct combination reaction of phosphorus pentoxide with water gives _______.
Explain the following reaction with their balanced chemical equation.
Ammonia gas reacts with hydrogen chloride.
Which changes occur during chemical changes?
The number of products formed in a synthesis reaction is ______
Define a combination reaction. Give one example of an exothermic combination reaction.
Which of the following are exothermic processes?
(i) Reaction of water with quick lime
(ii) Dilution of an acid
(iii) Evaporation of water
(iv) Sublimation of camphor (crystals)
Which of the following is not a physical change?
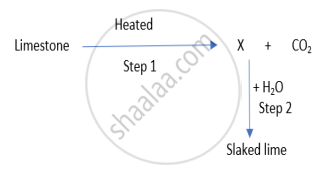
Identify the correct option from the given table which represents the type of reactions occurring in step 1 and step 2.
Balance the following chemical equation and identify the type of chemical reaction.
`"Mg"("s") + "Cl"_2("g") -> "MgCl"_2("s")`
Balance the following chemical equation and identify the type of chemical reaction.
`"Na"("s") + "S"("s") overset("(Fuse)")(->) "Na"_2"S"("s")`
Balance the following chemical equation and identify the type of chemical reaction.
`"CaO"("s") + "SiO"_2("s") -> "CaSio"_3("s")`
Give the chemical reaction when a few pieces of slaked lime are added to a beaker half-filled with water. Will it be an endothermic or exothermic reaction?
Read the text below and answer the questions that follow:
A small amount of hydrochloric acid was taken in a test tube. The test tube was heated. A glass rod was dipped in the ammonia solution and held on the top of the test tube. A white smoke was seen emanating from the tip of the glass rod.
- What must have happened?
- Which colour of gas is formed?
- Write the chemical equation for the reaction.
